Effect of Sarizotan, a 5-HT1a and D2-like receptor agonist, on respiration in three mouse models of Rett syndrome
- PMID: 24351104
- PMCID: PMC4068914
- DOI: 10.1165/rcmb.2013-0372OC
Effect of Sarizotan, a 5-HT1a and D2-like receptor agonist, on respiration in three mouse models of Rett syndrome
Abstract
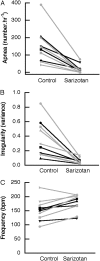
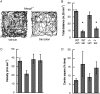
Disturbances in respiration are common and debilitating features of Rett syndrome (RTT). A previous study showed that the 5-HT1a receptor agonist (R)-(+)-8-hydroxy-dipropyl-2-aminotetralin hydrobromide (8-OH-DPAT) significantly reduced the incidence of apnea and the irregular breathing pattern in a mouse model of the disorder. 8-OH-DPAT, however, is not available for clinical practice. Sarizotan, a full 5-HT1a agonist and a dopamine D2-like agonist/partial agonist, has been used in clinical trials for the treatment of l-dopa-induced dyskinesia. The purpose of this study was to evaluate the effects of sarizotan on respiration and locomotion in mouse models of RTT. Studies were performed in Bird and Jaenisch strains of methyl-CpG-binding protein 2--deficient heterozygous female and Jaenisch strain Mecp2 null male mice and in knock-in heterozygous female mice of a common nonsense mutation (R168X). Respiratory pattern was determined with body plethysmography, and locomotion was determined with open-field recording. Sarizotan or vehicle was administered 20 minutes before a 30-minute recording of respiratory pattern or motor behavior. In separate studies, a crossover design was used to administer the drug for 7 and for 14 days. Sarizotan reduced the incidence of apnea in all three RTT mouse models to approximately 15% of their pretreatment levels. The irregular breathing pattern was corrected to that of wild-type littermates. When administered for 7 or 14 days, apnea decreased to 25 to 33% of the incidence seen with vehicle. This study indicates that the clinically approved drug sarizotan is an effective treatment for respiratory disorders in mouse models of RTT.
Figures





References
-
- Bird A. The methyl-CpG-binding protein MeCP2 and neurological disease. Biochem Soc Trans. 2008;36:575–583. - PubMed
-
- Chahrour M, Zoghbi HY. The story of Rett syndrome: from clinic to neurobiology. Neuron. 2007;56:422–437. - PubMed
-
- Weese-Mayer DE, Lieske SP, Boothby CM, Kenny AS, Bennett HL, Silvestri JM, Ramirez JM. Autonomic nervous system dysregulation: breathing and heart rate perturbation during wakefulness in young girls with Rett syndrome. Pediatr Res. 2006;60:443–449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

