The preparation of capsaicin-chitosan microspheres (CCMS) enteric coated tablets
- PMID: 24351818
- PMCID: PMC3876112
- DOI: 10.3390/ijms141224305
The preparation of capsaicin-chitosan microspheres (CCMS) enteric coated tablets
Abstract
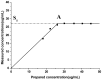
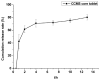
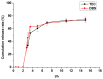
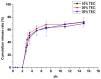
This study aimed to research the preparation and content determination of capsaicin-chitosan microspheres (CCMS) enteric coated tablets. The core tablets were prepared with the method of wet granulation. Nine formulae were designed to determine the optimal formula of the core tablet. Eudragit L100 was used to prepare the CCMS enteric-coated tablets. The effect of enteric coated formulation variables such as content of talc (10%, 25% and 40%), plasticisers (TEC and DBS), dosage of plasticiser (10%, 20% and 30%) and coating weight (2%, 3% and 5%) were evaluated for drug release characteristics. The in vitro release was studied using 0.1 N HCl and pH 6.8 phosphate buffer. Enteric coated tablets without ruptures or swelling behaviour over 2 h in 0.1 N HCl indicated that these tablets showed acid resistance. The accumulated release rate in phosphate buffer (pH 6.8) revealed that the prepared tablets were able to sustain drug release into the intestine and a first-order release was obtained for capsaicin. This research is the first report of the preparation and content determination of CCMS enteric coated tablets. The sustained release behavior of enteric coated formulations in pH 6.8 phosphate buffer demonstrated that it would be a potential drug delivery platform for sustained delivery of gastric irritant drugs.
Figures


Similar articles
-
Development of an enteric coating formulation and process for tablets primarily composed of a highly water-soluble, organic acid.Eur J Pharm Biopharm. 2001 Jan;51(1):71-6. doi: 10.1016/s0939-6411(00)00129-6. Eur J Pharm Biopharm. 2001. PMID: 11154906
-
Eudragit coating of chitosan-prednisolone conjugate microspheres and in vitro evaluation of coated microspheres.Drug Dev Ind Pharm. 2007 Aug;33(8):848-54. doi: 10.1080/03639040701377904. Drug Dev Ind Pharm. 2007. PMID: 17729102
-
Novel preparation of enteric-coated chitosan-prednisolone conjugate microspheres and in vitro evaluation of their potential as a colonic delivery system.Eur J Pharm Biopharm. 2008 Feb;68(2):260-6. doi: 10.1016/j.ejpb.2007.06.016. Epub 2007 Jul 4. Eur J Pharm Biopharm. 2008. PMID: 17703928
-
A Review on Enteric Coated Pellets Composed of Core Pellets Prepared by Extrusion-Spheronization.Recent Pat Drug Deliv Formul. 2019;13(2):83-90. doi: 10.2174/1872211313666190212115139. Recent Pat Drug Deliv Formul. 2019. PMID: 30747090 Review.
-
Preparation of the traditional Chinese medicine compound recipe heart-protecting musk pH-dependent gradient-release pellets.Drug Dev Ind Pharm. 2002 Nov;28(10):1261-73. doi: 10.1081/ddc-120015360. Drug Dev Ind Pharm. 2002. PMID: 12476872 Review.
Cited by
-
Synthesis and characterization of a polyurethane carrier used for a prolonged transmembrane transfer of a chili pepper extract.Int J Nanomedicine. 2018 Nov 6;13:7155-7166. doi: 10.2147/IJN.S181667. eCollection 2018. Int J Nanomedicine. 2018. PMID: 30464465 Free PMC article.
-
Synthesis of Chitosan Oligosaccharide-Loaded Glycyrrhetinic Acid Functionalized Mesoporous Silica Nanoparticles and In Vitro Verification of the Treatment of APAP-Induced Liver Injury.Molecules. 2023 May 17;28(10):4147. doi: 10.3390/molecules28104147. Molecules. 2023. PMID: 37241887 Free PMC article.
-
Microencapsulation of Capsaicin in Chitosan Microcapsules: Characterization, Release Behavior, and Pesticidal Properties against Tribolium castaneum (Herbst).Insects. 2022 Dec 27;14(1):27. doi: 10.3390/insects14010027. Insects. 2022. PMID: 36661955 Free PMC article.
-
Complex Relationship between Obesity and the Fat Mass and Obesity Locus.Int J Biol Sci. 2017 May 15;13(5):615-629. doi: 10.7150/ijbs.17051. eCollection 2017. Int J Biol Sci. 2017. PMID: 28539834 Free PMC article. Review.
-
The effects of COST on the differentiation of 3T3-L1 preadipocytes and the mechanism of action.Saudi J Biol Sci. 2017 Feb;24(2):251-255. doi: 10.1016/j.sjbs.2016.09.008. Epub 2016 Sep 10. Saudi J Biol Sci. 2017. PMID: 28149159 Free PMC article.
References
-
- Jequier E. Pathways to obesity. Int. J. Obes. Relat. Metab. Disord. 2002;26:12–17. - PubMed
-
- Cecchini M., Sassi F., Lauer J.A., Lee Y.Y., Guajardo-Barron V., Chisholm D. Tackling of unhealthy diets, physical inactivity, and obesity: Health effects and cost-effectiveness. Lancet. 2010;376:1775–1784. - PubMed
-
- Cope M.B., Allison D.B. Obesity: person and population. Obesity. 2006;14:156S–159S. - PubMed
-
- Orsi C.M., Hale D.E., Lynch J.L. Pediatric obesity epidemiology. Curr. Opin. Endocrinol. Diabetes Obes. 2011;18:14–22. - PubMed
-
- Birch L.L., Ventura A.K. Preventing childhood obesity: What works? Int. J. Obes. 2009;33:S74–S81. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

