Stereocontrolled synthesis and functionalization of cyclobutanes and cyclobutanones
- PMID: 24352013
- PMCID: PMC6269998
- DOI: 10.3390/molecules181215541
Stereocontrolled synthesis and functionalization of cyclobutanes and cyclobutanones
Abstract
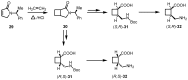
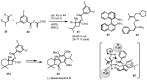
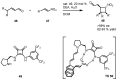
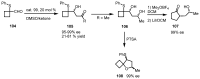
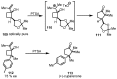
In the last decade a certain number of new cyclobutane and cyclobutanone synthesis and functionalization protocols have been published. Organo- and biocatalyzed eco-friendly approaches to cyclobutane-containing molecules have been developed with interesting results. Also, successful new total synthesis of bioactive compounds and drugs have been recently reported where a four membered ring represented the key intermediate. Therefore, the rising interest in this field represents a great point of discussion for the scientific community, disclosing the synthetic potential of strained four membered ring carbocyclic compounds. Herein we report a critical survey on the literature concerning the enantiocontrolled synthesis and functionalization of cyclobutane derivatives, with particular attention to metal-free, low impact methodologies, published during the period 2000-2013.
Figures



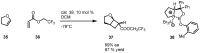








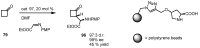
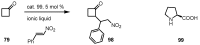





Similar articles
-
Scopariusicides, Novel Unsymmetrical Cyclobutanes: Structural Elucidation and Concise Synthesis by a Combination of Intermolecular [2 + 2] Cycloaddition and C-H Functionalization.Org Lett. 2015 Dec 18;17(24):6062-5. doi: 10.1021/acs.orglett.5b03079. Epub 2015 Nov 29. Org Lett. 2015. PMID: 26617269
-
Cobalt Catalysis for Enantioselective Cyclobutanone Construction.J Am Chem Soc. 2017 Aug 2;139(30):10208-10211. doi: 10.1021/jacs.7b05327. Epub 2017 Jul 13. J Am Chem Soc. 2017. PMID: 28704053 Free PMC article.
-
Cyclobutane amino acid analogues of furanomycin obtained by a formal [2 + 2] cycloaddition strategy promoted by methylaluminoxane.J Org Chem. 2010 Feb 5;75(3):545-52. doi: 10.1021/jo9025258. J Org Chem. 2010. PMID: 20038109
-
Advances in the enantioselective synthesis of carbocyclic nucleosides.Chem Soc Rev. 2013 Jun 21;42(12):5056-72. doi: 10.1039/c3cs00003f. Chem Soc Rev. 2013. PMID: 23471263 Review.
-
[2 + 2]-Cycloaddition-derived cyclobutane natural products: structural diversity, sources, bioactivities, and biomimetic syntheses.Nat Prod Rep. 2023 Jun 21;40(6):1094-1129. doi: 10.1039/d2np00034b. Nat Prod Rep. 2023. PMID: 36314443 Review.
Cited by
-
Enantioselective C-H Arylation and Vinylation of Cyclobutyl Carboxylic Amides.ACS Catal. 2018 Mar 2;8(3):2577-2584. doi: 10.1021/acscatal.8b00069. Epub 2018 Feb 5. ACS Catal. 2018. PMID: 29531850 Free PMC article.
-
Effect of Reaction Media on Photosensitized [2+2]-Cycloaddition of Cinnamates.ChemistryOpen. 2020 May 5;9(6):649-656. doi: 10.1002/open.202000092. eCollection 2020 Jun. ChemistryOpen. 2020. PMID: 32499991 Free PMC article.
-
Tandem catalysis for asymmetric coupling of ethylene and enynes to functionalized cyclobutanes.Science. 2018 Jul 6;361(6397):68-72. doi: 10.1126/science.aat6205. Science. 2018. PMID: 29976822 Free PMC article.
-
PdII -Catalyzed Enantioselective C(sp3 )-H Activation/Cross-Coupling Reactions of Free Carboxylic Acids.Angew Chem Int Ed Engl. 2019 Feb 11;58(7):2134-2138. doi: 10.1002/anie.201813055. Epub 2019 Jan 16. Angew Chem Int Ed Engl. 2019. PMID: 30536546 Free PMC article.
-
Stereospecific Synthesis of Cyclobutanones and Spirohexanones via Formal [3 + 1] Cycloaddition of Cyclopropanones with Sulfur Ylides.Org Lett. 2025 Aug 1;27(30):8332-8337. doi: 10.1021/acs.orglett.5c02601. Epub 2025 Jul 22. Org Lett. 2025. PMID: 40693674 Free PMC article.
References
-
- Rappaport Z., Liebman J.F. The Chemistry of Cyclobutanes Part I. Wiley; Chichester, UK: 2005.
-
- Dabrowski J.A., Moebius D.C., Wommack A.J., Kornahrens A.F., Kingsbury J.S. Catalytic and regioselective ring expansion of arylcyclobutanones with trimethylsilyldiazomethane. Ligand-dependent entry to β-ketosilane or enolsilane adducts. Org. Lett. 2010;12:3598–3601. doi: 10.1021/ol101136a. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

