PDK1 selectively phosphorylates Thr(308) on Akt and contributes to human platelet functional responses
- PMID: 24352480
- PMCID: PMC4079046
- DOI: 10.1160/TH13-06-0484
PDK1 selectively phosphorylates Thr(308) on Akt and contributes to human platelet functional responses
Abstract
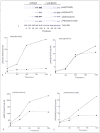
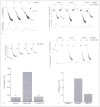
3-phosphoinositide-dependent protein kinase 1 (PDK1), a member of the protein A,G and C (AGC) family of proteins, is a Ser/Thr protein kinase that can phosphorylate and activate other protein kinases from the AGC family, including Akt at Thr308, all of which play important roles in mediating cellular responses. The functional role of PDK1 or the importance of phosphorylation of Akt on Thr308 for its activity has not been investigated in human platelets. In this study, we tested two pharmacological inhibitors of PDK1, BX795 and BX912, to assess the role of Thr308 phosphorylation on Akt. PAR4-induced phosphorylation of Akt on Thr308 was inhibited by BX795 without affecting phosphorylation of Akt on Ser473. The lack of Thr308 phosphorylation on Akt also led to the inhibition of PAR4-induced phosphorylation of two downstream substrates of Akt, viz. GSK3β and PRAS40. In vitro kinase activity of Akt was completely abolished if Thr308 on Akt was not phosphorylated. BX795 caused inhibition of 2-MeSADP-induced or collagen-induced aggregation, ATP secretion and thromboxane generation. Primary aggregation induced by 2-MeSADP was also inhibited in the presence of BX795. PDK1 inhibition also resulted in reduced clot retraction indicating its role in outside-in signalling. These results demonstrate that PDK1 selectively phosphorylates Thr308 on Akt thereby regulating its activity and plays a positive regulatory role in platelet physiological responses.
Keywords: Kinases; platelet pharmacology; signal transduction.
Conflict of interest statement
None declared.
Figures




Similar articles
-
PDK1 governs thromboxane generation and thrombosis in platelets by regulating activation of Raf1 in the MAPK pathway.J Thromb Haemost. 2018 Jun;16(6):1211-1225. doi: 10.1111/jth.14005. Epub 2018 May 8. J Thromb Haemost. 2018. PMID: 29575487 Free PMC article.
-
PDK1 regulates platelet activation and arterial thrombosis.Blood. 2013 May 2;121(18):3718-26. doi: 10.1182/blood-2012-10-461897. Epub 2013 Feb 26. Blood. 2013. PMID: 23444402
-
ADP-stimulated activation of Akt during integrin outside-in signaling promotes platelet spreading by inhibiting glycogen synthase kinase-3β.Arterioscler Thromb Vasc Biol. 2012 Sep;32(9):2232-40. doi: 10.1161/ATVBAHA.112.254680. Epub 2012 Jul 19. Arterioscler Thromb Vasc Biol. 2012. PMID: 22814751 Free PMC article.
-
Regulation of protein kinases in insulin, growth factor and Wnt signalling.Curr Opin Struct Biol. 2002 Dec;12(6):761-7. doi: 10.1016/s0959-440x(02)00386-x. Curr Opin Struct Biol. 2002. PMID: 12504681 Review.
-
Acute regulation of PDK1 by a complex interplay of molecular switches.Biochem Soc Trans. 2014 Oct;42(5):1435-40. doi: 10.1042/BST20140222. Biochem Soc Trans. 2014. PMID: 25233428 Review.
Cited by
-
PTEN-GSK3β-MOB1 axis controls neurite outgrowth in vitro and in vivo.Cell Mol Life Sci. 2018 Dec;75(23):4445-4464. doi: 10.1007/s00018-018-2890-0. Epub 2018 Aug 1. Cell Mol Life Sci. 2018. PMID: 30069702 Free PMC article.
-
Reactive Oxygen Species: A Double-Edged Sword in the Modulation of Cancer Signaling Pathway Dynamics.Cells. 2025 Aug 6;14(15):1207. doi: 10.3390/cells14151207. Cells. 2025. PMID: 40801639 Free PMC article. Review.
-
EMC2 promotes breast cancer progression and enhances sensitivity to PDK1/AKT inhibition by deubiquitinating ENO1.Int J Biol Sci. 2025 Mar 24;21(6):2629-2646. doi: 10.7150/ijbs.109192. eCollection 2025. Int J Biol Sci. 2025. PMID: 40303285 Free PMC article.
-
The Influence of Circadian Rhythms on DNA Damage Repair in Skin Photoaging.Int J Mol Sci. 2024 Oct 11;25(20):10926. doi: 10.3390/ijms252010926. Int J Mol Sci. 2024. PMID: 39456709 Free PMC article. Review.
-
Medulloblastoma and the DNA Damage Response.Front Oncol. 2022 Jun 7;12:903830. doi: 10.3389/fonc.2022.903830. eCollection 2022. Front Oncol. 2022. PMID: 35747808 Free PMC article. Review.
References
-
- Brass LF. Thrombin and platelet activation. Chest. 2003;124:18S–25S. - PubMed
-
- Kroll MH, Hellums JD, McIntire LV, et al. Platelets and shear stress. Blood. 1996;88:1525–1541. - PubMed
-
- Savage B, Saldivar E, Ruggeri ZM. Initiation of platelet adhesion by arrest onto fibrinogen or translocation on von Willebrand factor. Cell. 1996;84:289–97. - PubMed
-
- Nieswandt B, Watson SP. Platelet-collagen interaction: is GPVI the central receptor? Blood. 2003;102:449–461. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

