Impact of proficiency testing program for laboratories conducting early diagnosis of HIV-1 infection in infants in low- to middle-income countries
- PMID: 24353004
- PMCID: PMC3957753
- DOI: 10.1128/JCM.03097-13
Impact of proficiency testing program for laboratories conducting early diagnosis of HIV-1 infection in infants in low- to middle-income countries
Abstract
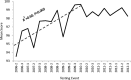
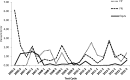
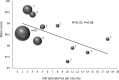
A voluntary, cost-free external quality assessment (EQA) program established by the U.S. Centers for Disease Control and Prevention (CDC) was implemented to primarily monitor the performance of laboratories conducting HIV Early Infant Diagnosis (EID) from dried blood spots (DBS) in low- to middle-income countries since 2006. Ten blind DBS proficiency test (PT) specimens and 100 known HIV-positive and -negative DBS specimens (to be used as internal controls) were shipped triannually to participating laboratories with reports for the PT specimens due within 30 days. The participant's results and a summary of the performance of all participating laboratories and each diagnostic method were provided after each test cycle. Enrollment in the CDC PT program expanded progressively from 17 laboratories from 11 countries in 2006 to include 136 laboratories from 41 countries at the end of 2012. Despite external pressures to test and treat more children while expanding EID programs, mean PT test scores significantly improved over time as demonstrated by the upward trend from mid-2006 to the end of 2012 (P=0.001) and the increase in the percentage of laboratories scoring 100% (P=0.003). The mean test scores plateaued over the past 10 testing cycles, ranging between 98.2% and 99.7%, and discordant test results still occur but at a rate of no higher than 2.6%. Analysis of these test results suggests a positive impact of proficiency testing on the testing performance of the participating laboratories, and a continuous training program and proficiency testing participation may translate into laboratories improving their testing accuracy.
Figures

Similar articles
-
Monitoring the quality of HIV-1 viral load testing through a proficiency testing program using dried tube specimens in resource-limited settings.J Clin Microbiol. 2015 Apr;53(4):1129-36. doi: 10.1128/JCM.02780-14. Epub 2015 Jan 21. J Clin Microbiol. 2015. PMID: 25609733 Free PMC article.
-
Development of a proficiency testing program for the HIV-1 BED incidence assay in China.Sci Rep. 2014 Mar 28;4:4512. doi: 10.1038/srep04512. Sci Rep. 2014. PMID: 24676229 Free PMC article.
-
A comprehensive evaluation of the proficiency testing program for the HIV-1 BED incidence assay.J Clin Microbiol. 2011 Oct;49(10):3470-3. doi: 10.1128/JCM.01122-11. Epub 2011 Aug 10. J Clin Microbiol. 2011. PMID: 21832016 Free PMC article.
-
Proficiency testing/external quality assessment: current challenges and future directions.Clin Chem. 2011 Dec;57(12):1670-80. doi: 10.1373/clinchem.2011.168641. Epub 2011 Sep 30. Clin Chem. 2011. PMID: 21965556 Review.
-
Implementation of Good Clinical Laboratory Practice (GCLP) guidelines within the External Quality Assurance Program Oversight Laboratory (EQAPOL).J Immunol Methods. 2014 Jul;409:91-8. doi: 10.1016/j.jim.2013.09.012. Epub 2013 Oct 9. J Immunol Methods. 2014. PMID: 24120573 Free PMC article. Review.
Cited by
-
Validation of the point-of-care (POC) technologies Xpert HIV-1 Qual and m-PIMA HIV 1/2 detect for early diagnosis of HIV-1 and HIV-2 in Senegal.Pan Afr Med J. 2022 Sep 27;43:42. doi: 10.11604/pamj.2022.43.42.32714. eCollection 2022. Pan Afr Med J. 2022. PMID: 36523274 Free PMC article.
-
Diagnosis of Human Immunodeficiency Virus Infection.Clin Microbiol Rev. 2018 Nov 28;32(1):e00064-18. doi: 10.1128/CMR.00064-18. Print 2019 Jan. Clin Microbiol Rev. 2018. PMID: 30487166 Free PMC article. Review.
-
Impact of External Quality Assurance on the Quality of Xpert MTB/RIF Testing in Viet Nam.J Clin Microbiol. 2019 Feb 27;57(3):e01669-18. doi: 10.1128/JCM.01669-18. Print 2019 Mar. J Clin Microbiol. 2019. PMID: 30567748 Free PMC article.
-
Implementation of Novel Quality Assurance Program for Hepatitis C Viral Load Point of Care Testing.Viruses. 2022 Aug 30;14(9):1929. doi: 10.3390/v14091929. Viruses. 2022. PMID: 36146736 Free PMC article.
-
Quality assurance for HIV point-of-care testing and treatment monitoring assays.Afr J Lab Med. 2016 Oct 17;5(2):557. doi: 10.4102/ajlm.v5i2.557. eCollection 2016. Afr J Lab Med. 2016. PMID: 28879133 Free PMC article.
References
-
- UNAIDS 2010. UNAIDS report on the global AIDS epidemic. UNAIDS, Geneva, Switzerland
-
- Berk DR, Falkovitz-Halpern MS, Hill DW, Albin C, Arrieta A, Bork JM, Cohan D, Nilson B, Petru A, Ruiz J, Weintrub PS, Wenman W, Maldonado YA. 2005. Temporal trends in early clinical manifestations of perinatal HIV infection in a population-based cohort. JAMA 293:2221–2231. 10.1001/jama.293.18.2221 - DOI - PubMed
-
- WHO 2010. Antiretroviral therapy for HIV infection in infants and children: towards universal access. Recommendations for a public health approach. World Health Organization, Geneva, Switzerland - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

