Gsta 4 Null Mouse Embryonic Fibroblasts Exhibit Enhanced Sensitivity to Oxidants: Role of 4-Hydroxynonenal in Oxidant Toxicity
- PMID: 24353929
- PMCID: PMC3864047
- DOI: 10.4236/ojapo.2013.21001
Gsta 4 Null Mouse Embryonic Fibroblasts Exhibit Enhanced Sensitivity to Oxidants: Role of 4-Hydroxynonenal in Oxidant Toxicity
Abstract
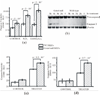
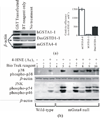
The alpha class glutathione s-transferase (GST) isozyme GSTA4-4 (EC2.5.1.18) exhibits high catalytic efficiency to-wards 4-hydroxynon-2-enal (4-HNE), a major end product of oxidative stress induced lipid peroxidation. Exposure of cells and tissues to heat, radiation, and chemicals has been shown to induce oxidative stress resulting in elevated concentrations of 4-HNE that can be detrimental to cell survival. Alternatively, at physiological levels 4-HNE acts as a signaling molecule conveying the occurrence of oxidative events initiating the activation of adaptive pathways. To examine the impact of oxidative/electrophilic stress in a model with impaired 4-HNE metabolizing capability, we disrupted the Gsta4 gene that encodes GSTA4-4 in mice. The effect of electrophile and oxidants on embryonic fibroblasts (MEF) isolated from wild type (WT) and Gsta4 null mice were examined. Results indicate that in the absence of GSTA4-4, oxidant-induced toxicity is potentiated and correlates with elevated accumulation of 4-HNE adducts and DNA damage. Treatment of Gsta4 null MEF with 1,1,4-tris(acetyloxy)-2(E)-nonene [4-HNE(Ac)3], a pro-drug form of 4-HNE, resulted in the activation and phosphorylation of the c-jun-N-terminal kinase (JNK), extracellular-signal-regulated kinases (ERK 1/2) and p38 mitogen activated protein kinases (p38 MAPK) accompanied by enhanced cleavage of caspase-3. Interestingly, when recombinant mammalian or invertebrate GSTs were delivered to Gsta4 null MEF, activation of stress-related kinases in 4-HNE(Ac)3 treated Gsta4 null MEF were inversely correlated with the catalytic efficiency of delivered GSTs towards 4-HNE. Our data suggest that GSTA4-4 plays a major role in protecting cells from the toxic effects of oxidant chemicals by attenuating the accumulation of 4-HNE.
Keywords: Glutathione Transferase; Lipid Peroxidation; Oxidants; Protein Adducts; Stress Kinases.
Conflict of interest statement
Conflicts of interest: The authors report no conflicts of interest and are responsible for the content and writing of the paper.
Figures





Similar articles
-
Loss of glutathione S-transferase A4 accelerates obstruction-induced tubule damage and renal fibrosis.J Pathol. 2012 Dec;228(4):448-58. doi: 10.1002/path.4067. Epub 2012 Jul 26. J Pathol. 2012. PMID: 22711583 Free PMC article.
-
Protection from oxidative and electrophilic stress in the Gsta4-null mouse heart.Cardiovasc Toxicol. 2013 Dec;13(4):347-56. doi: 10.1007/s12012-013-9215-1. Cardiovasc Toxicol. 2013. PMID: 23690225 Free PMC article.
-
Effects of Local Heart Irradiation in a Glutathione S-Transferase Alpha 4-Null Mouse Model.Radiat Res. 2015 Jun;183(6):610-9. doi: 10.1667/RR13979.1. Epub 2015 May 26. Radiat Res. 2015. PMID: 26010708 Free PMC article.
-
Lipid peroxidation and cell cycle signaling: 4-hydroxynonenal, a key molecule in stress mediated signaling.Acta Biochim Pol. 2003;50(2):319-36. Acta Biochim Pol. 2003. PMID: 12833161 Review.
-
Antioxidant role of glutathione S-transferases: 4-Hydroxynonenal, a key molecule in stress-mediated signaling.Toxicol Appl Pharmacol. 2015 Dec 15;289(3):361-70. doi: 10.1016/j.taap.2015.10.006. Epub 2015 Oct 23. Toxicol Appl Pharmacol. 2015. PMID: 26476300 Free PMC article. Review.
Cited by
-
Cox4i2 Triggers an Increase in Reactive Oxygen Species, Leading to Ferroptosis and Apoptosis in HHV7 Infected Schwann Cells.Front Mol Biosci. 2021 May 7;8:660072. doi: 10.3389/fmolb.2021.660072. eCollection 2021. Front Mol Biosci. 2021. PMID: 34026834 Free PMC article.
-
The Multifaceted Regulation of Mitochondria in Ferroptosis.Life (Basel). 2021 Mar 10;11(3):222. doi: 10.3390/life11030222. Life (Basel). 2021. PMID: 33801920 Free PMC article. Review.
-
Redox Imbalance and Viral Infections in Neurodegenerative Diseases.Oxid Med Cell Longev. 2016;2016:6547248. doi: 10.1155/2016/6547248. Epub 2016 Mar 27. Oxid Med Cell Longev. 2016. PMID: 27110325 Free PMC article. Review.
-
IRGM/Irgm1 facilitates macrophage apoptosis through ROS generation and MAPK signal transduction: Irgm1+/- mice display increases atherosclerotic plaque stability.Theranostics. 2021 Sep 9;11(19):9358-9375. doi: 10.7150/thno.62797. eCollection 2021. Theranostics. 2021. PMID: 34646375 Free PMC article.
-
Mechanisms and cross-talk of regulated cell death and their epigenetic modifications in tumor progression.Mol Cancer. 2024 Nov 29;23(1):267. doi: 10.1186/s12943-024-02172-y. Mol Cancer. 2024. PMID: 39614268 Free PMC article. Review.
References
-
- Winczura A, Zdzalik D, Tudek B. Damage of DNA and Proteins by Major Lipid Peroxidation Products in Genome Stability. Free Radical Research. 2012;Vol. 46(No. 4):442–459. - PubMed
-
- Voulgaridou GP, Anestopoulos I, Franco R, Panayiotidis MI, Pappa A. DNA Damage Induced by Endogenous Aldehydes: Current State of Knowledge. Mutation Research. 2011;Vol. 711(No. 1–2):13–27. - PubMed
-
- Esterbauer H, Schaur RJ, Zollner H. Chemistry and Biochemistry of 4-Hydroxynonenal, Malonaldehyde and Related Aldehydes. Free Radical Biology and Medicine. 1991;Vol. 11(No. 1):81–128. - PubMed
-
- Singh SP, Chen T, Chen L, Mei N, McLain E, Samokyszyn V, et al. Mutagenic Effects of 4-Hydroxy-nonenal Triacetate, a Chemically Protected form of the Lipid Peroxidation Product 4-Hydroxynonenal, as Assayed in L5178Y/Tk+/-Mouse Lymphoma Cells. Journal of Pharmacology and Experimental Therapeutics. 2005;Vol. 313(No. 2):855–861. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
