Triptans for acute cluster headache
- PMID: 24353996
- PMCID: PMC6494511
- DOI: 10.1002/14651858.CD008042.pub3
Triptans for acute cluster headache
Abstract
Background: This is an updated version of the original Cochrane review published in Issue 4, 2010 (Law 2010). Cluster headache is an uncommon,severely painful, and disabling condition, with rapid onset. Validated treatment options are limited; first-line therapy includes inhaled oxygen. Other therapies such as intranasal lignocaine and ergotamine are not as commonly used and are less well studied. Triptans are successfully used to treat migraine attacks and they may also be useful for cluster headache.
Objectives: To assess the efficacy and tolerability of the triptan class of drugs compared to placebo and other active interventions in the acute treatment of episodic and chronic cluster headache in adult patients.
Search methods: We searched the Cochrane Central Register of Controlled Trials (CENTRAL),MEDLINE, EMBASE, ClinicalTrials.gov, and reference lists for studies from inception to 22 January 2010 for the original review, and from 2009 to 4 April 2013 for this update.
Selection criteria: Randomised, double-blind, placebo-controlled studies of triptans for acute treatment of cluster headache episodes.
Data collection and analysis: Two review authors independently assessed study quality and extracted data. Numbers of participants with different levels of pain relief,requiring rescue medication, and experiencing adverse events and headache-associated symptoms in treatment and control groups were used to calculate relative risk and numbers needed to treat for benefit (NNT) and harm (NNH).
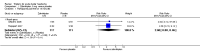
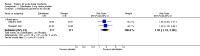
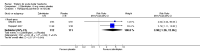
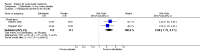
Main results: New searches in 2013 did not identify any relevant new studies.All six included studies used a single dose of triptan to treat an attack of moderate to severe pain intensity. Subcutaneous sumatriptan was given to 131 participants at a 6 mg dose, and 88 at a 12 mg dose. Oral or intranasal zolmitriptan was given to 231 participants ata 5 mg dose, and 223 at a 10 mg dose. Placebo was given to 326 participants.Triptans were more effective than placebo for headache relief and pain-free responses. By 15 minutes after treatment with subcutaneous sumatriptan 6 mg, 48% of participants were pain-free and 75% had no pain or mild pain (17% and 32% respectively with placebo).NNTs for subcutaneous sumatriptan 6 mg were 3.3 (95% CI 2.4 to 5.0) and 2.4 (1.9 to 3.2) respectively. Intranasal zolmitriptan 10mg was of less benefit, with 12% of participants pain-free and 28% with no or mild pain (3% and 7% respectively with placebo).NNTs for intranasal zolmitriptan 10 mg were 11 (6.4 to 49) and 4.9 (3.3 to 9.2) respectively.
Authors' conclusions: Based on limited data, subcutaneous sumatriptan 6 mg was superior to intranasal zolmitriptan 5 mg or 10 mg for rapid (15 minute)responses, which are important in this condition. Oral routes of administration are not appropriate.
Conflict of interest statement
RAM has consulted for various pharmaceutical companies. RAM has received lecture fees from pharmaceutical companies, related to analgesics and other healthcare interventions. RAM and SD have received research support from charities, government and industry sources at various times. Support for this review was from the Oxford Pain Research Trust, the NHS Cochrane Collaboration Programme Grant Scheme, and the NIHR Biomedical Research Centre Programme. None had any input into the review at any stage. SL has no interests to declare.
Figures


















Update of
-
Triptans for acute cluster headache.Cochrane Database Syst Rev. 2010 Apr 14;(4):CD008042. doi: 10.1002/14651858.CD008042.pub2. Cochrane Database Syst Rev. 2010. Update in: Cochrane Database Syst Rev. 2013 Jul 17;(7):CD008042. doi: 10.1002/14651858.CD008042.pub3. PMID: 20393964 Free PMC article. Updated.
References
References to studies included in this review
Bahra 2000 {published data only}
-
- Bahra A, Gawel MJ, Hardebo JE, Millson D, Breen SA, Goadsby PJ. Oral zolmitriptan is effective in the acute treatment of cluster headache. Neurology 2000;54(9):1832‐9. - PubMed
Cittadini 2006 {published data only}
Ekbom 1991 {published data only}
-
- The Sumatriptan Cluster Headache Study Group. Treatment of acute cluster headache with sumatriptan. New England Journal of Medicine 1991;325(5):322‐6. - PubMed
Ekbom 1993 {published data only}
-
- Ekbom K, Monstad I, Prusinski A, Cole JA, Pilgrim AJ, Noronha D. Subcutaneous sumatriptan in the acute treatment of cluster headache: a dose comparison study. The Sumatriptan Cluster Headache Study Group. Acta Neurologica Scandinavica 1993;88(1):63‐9. - PubMed
Rapoport 2007 {published data only}
Van Vliet 2003 {published data only}
-
- Vliet JA, Bahra A, Martin V, Ramadan N, Aurora SK, Mathew NT, et al. Intranasal sumatriptan in cluster headache: randomized placebo‐controlled double‐blind study. Neurology 2003;60(4):630‐3. - PubMed
References to studies excluded from this review
Goadsby 1994 {published data only}
-
- Goadsby PJ. The clinical profile of sumatriptan: cluster headache. European Neurolology 1994;34 Suppl 2:35‐9. - PubMed
Hardebo 1998 {published data only}
-
- Hardebo JE, Dahlöf C. Sumatriptan nasal spray (20 mg/dose) in the acute treatment of cluster headache. Cephalalgia 1998;18(7):487‐9. - PubMed
Additional references
Bennett 2008
Cluster Headache Study Group 1991
-
- The Sumatriptan Cluster Headache Study Group. Treatment of acute cluster headache with sumatriptan. New England Journal of Medicine 1991;325(5):322‐6. - PubMed
Cohen 2009
-
- Cohen A, Burns B, Goadsby P. High flow oxygen for the treatment of cluster headache: a randomised, placebo controlled trial. JAMA 2009;302(22):2451‐7. - PubMed
Cook 1995
D'Alessandro 1986
-
- D'Alessandro R, Gamberini G, Benassi G, Morganti G, Cortelli, Lugaresi E. Cluster headache in the Republic of San Marino. Cephalalgia 1986;6(3):159‐62. - PubMed
Derry 2009
Dodick 2004
-
- Dodick DW, Martin VT, Smith T, Silberstein S. Cardiovascular tolerability and safety of triptans: a review of clinical data. Headache 2004;44 Suppl 1:S20‐30. - PubMed
Ekbom 1978
-
- Ekbom K, Ahlborg B, Schéle R. Prevalence of migraine and cluster headache in Swedish men of 18. Headache 1978;18(1):9‐19. - PubMed
Elbourne 2002
Gobel 1998
-
- Göbel H, Lindner V, Heinze A, Ribbat M, Deuschl G. Acute therapy for cluster headache with sumatriptan: findings of a one‐year long‐term study. Neurology 1998;51(3):908‐11. - PubMed
Hedlund 2009
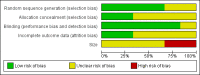
Higgins 2011
-
- Higgins JPT, Green S (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Altman DG, Sterne JAC editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
IHS 1988
-
- Headache Classification Committee of the International Headache Society. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia 1988;8 Suppl 7:1‐96. - PubMed
IHS 2004
-
- Headache Classification Subcommittee of the International Headache Society. The international classification of headache disorders. 2nd edition. Cephalalgia 2004;24 Suppl 1:1‐160. - PubMed
IHS Trial Guidelines 1995
-
- Lipton RB, Micieli G, Russell D, Solomon S, Tfelt‐Hansen P, Waldenlind E, for the International Headache Society Committee on Clinical Trials in Cluster Headache. Guidelines for controlled trials of drugs in cluster headache. Cephalalgia 1995;15(6):452‐62. - PubMed
Jadad 1996
-
- Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM, Gavaghan DJ, et al. Assessing the quality of reports of randomised controlled trials: is blinding necessary?. Controlled Clinical Trials 1996;17(1):1‐12. - PubMed
L'Abbé 1987
-
- L'Abbé KA, Detsky AS, O'Rourke K. Meta‐analysis in clinical research. Annals of Internal Medicine 1987;107(2):224‐33. - PubMed
May 1998
-
- May A, Bahra A, Büchel C, Frackowiak RS, Goadsby PJ. Hypothalamic activation in cluster headache attacks. Lancet 1998;352(9124):275‐8. - PubMed
McAlister 2001
-
- McAlister FA, Sackett DL. Active‐control equivalence trials and antihypertensive agents. American Journal of Medicine 2001;111(7):553‐8. - PubMed
Moore 1998
-
- Moore RA, Gavaghan D, Tramer MR, Collins SL, McQuay HJ. Size is everything ‐ large amounts of information are needed to overcome random effects in estimating direction and magnitude of treatment effects. Pain 1998;78(3):209‐16. - PubMed
Moore 2003
-
- Moore A, Edwards J, Barden J, McQuay H. Bandolier's little book of pain. Oxford: Oxford University Press, 2003.
Moore 2008
-
- Moore RA, Barden J, Derry S, McQuay HJ. Managing potential publication bias. In: McQuay HJ, Kalso E, Moore RA editor(s). Systematic Reviews in Pain Research: Methodology Refined. Seattle: IASP Press, 2008:15‐24. [ISBN: 978‐0‐931092‐69‐5]
Moore 2010
Morris 1995
-
- Morris JA, Gardner MJ. Calculating confidence intervals for relative risk, odds ratio and standardized ratios and rates. Gardner MJ, Altman DG, editors(s). Statistics with confidence ‐ confidence intervals and statistical guidelines. London: British Medical Journal, 1995:50‐63.
Nesbitt 2012
Plosker 1994
-
- Plosker GL, McTavish D. Sumatriptan. A reappraisal of its pharmacology and therapeutic efficacy in the acute treatment of migraine and cluster headache. Drugs 1994;47(4):622‐51. - PubMed
RevMan 2012 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.2. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2012.
Rozen 2011
Russell 2004
Sjöstrand 2010
Tepper 2003
-
- Tepper SJ, Millson D. Safety profile of the triptans. Expert Opinion on Drug Safety 2003;2(2):123‐32. - PubMed
Tfelt‐Hansen 2012
Torelli 2005
-
- Torelli P, Beghi E, Manzoni GC. Cluster headache prevalence in the Italian general population. Neurology 2005;64(3):469‐74. - PubMed
Tramèr 1997
Tyagi 2009
-
- Tyagi A, Matharu M. Evidence base for the medical treatments used in cluster headache. Current Pain and Headache Reports 2009;13(2):168‐78. - PubMed
Welch 2000
-
- Welch KM, Mathew NT, Stone P, Rosamond W, Saiers J, Gutterman D. Tolerability of sumatriptan: clinical trials and post‐marketing experience. Cephalalgia 2000;20(8):687‐95. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

