Atlas of coronavirus replicase structure
- PMID: 24355834
- PMCID: PMC7114488
- DOI: 10.1016/j.virusres.2013.12.004
Atlas of coronavirus replicase structure
Abstract
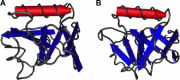
The international response to SARS-CoV has produced an outstanding number of protein structures in a very short time. This review summarizes the findings of functional and structural studies including those derived from cryoelectron microscopy, small angle X-ray scattering, NMR spectroscopy, and X-ray crystallography, and incorporates bioinformatics predictions where no structural data is available. Structures that shed light on the function and biological roles of the proteins in viral replication and pathogenesis are highlighted. The high percentage of novel protein folds identified among SARS-CoV proteins is discussed.
Keywords: Cryoelectron microscopy; NMR; Nonstructural protein; Protein structure; SARS coronavirus; Small angle X-ray scattering; X-ray crystallography.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures




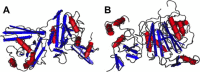
Similar articles
-
SARS-unique fold in the Rousettus bat coronavirus HKU9.Protein Sci. 2017 Sep;26(9):1726-1737. doi: 10.1002/pro.3208. Epub 2017 Jun 15. Protein Sci. 2017. PMID: 28580734 Free PMC article.
-
Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir.Science. 2020 Jun 26;368(6498):1499-1504. doi: 10.1126/science.abc1560. Epub 2020 May 1. Science. 2020. PMID: 32358203 Free PMC article.
-
NMR assignment of the domain 513-651 from the SARS-CoV nonstructural protein nsp3.Biomol NMR Assign. 2007 Dec;1(2):191-4. doi: 10.1007/s12104-007-9052-x. Epub 2007 Oct 30. Biomol NMR Assign. 2007. PMID: 19636862 Free PMC article.
-
The coronavirus replicase: insights into a sophisticated enzyme machinery.Adv Exp Med Biol. 2006;581:3-11. doi: 10.1007/978-0-387-33012-9_1. Adv Exp Med Biol. 2006. PMID: 17037497 Free PMC article. Review. No abstract available.
-
RNA structure analysis of alphacoronavirus terminal genome regions.Virus Res. 2014 Dec 19;194:76-89. doi: 10.1016/j.virusres.2014.10.001. Epub 2014 Oct 13. Virus Res. 2014. PMID: 25307890 Free PMC article. Review.
Cited by
-
Translational Control of COVID-19 and Its Therapeutic Implication.Front Immunol. 2022 Mar 29;13:857490. doi: 10.3389/fimmu.2022.857490. eCollection 2022. Front Immunol. 2022. PMID: 35422818 Free PMC article. Review.
-
Viral and cellular translation during SARS-CoV-2 infection.FEBS Open Bio. 2022 Sep;12(9):1584-1601. doi: 10.1002/2211-5463.13413. Epub 2022 Apr 25. FEBS Open Bio. 2022. PMID: 35429230 Free PMC article. Review.
-
1H, 13C and 15N backbone chemical shift assignments of SARS-CoV-2 nsp3a.Biomol NMR Assign. 2021 Apr;15(1):173-176. doi: 10.1007/s12104-020-10001-8. Epub 2021 Jan 21. Biomol NMR Assign. 2021. PMID: 33475934 Free PMC article.
-
Dissecting infectious bronchitis virus-induced host shutoff at the translation level.J Virol. 2024 Jul 23;98(7):e0083024. doi: 10.1128/jvi.00830-24. Epub 2024 Jun 28. J Virol. 2024. PMID: 38940559 Free PMC article.
-
Receptor tyrosine kinase inhibitors block proliferation of TGEV mainly through p38 mitogen-activated protein kinase pathways.Antiviral Res. 2020 Jan;173:104651. doi: 10.1016/j.antiviral.2019.104651. Epub 2019 Nov 18. Antiviral Res. 2020. PMID: 31751591 Free PMC article.
References
-
- Akaji K., Konno H., Mitsui H., Teruya K., Shimamoto Y., Hattori Y., Ozaki T., Kusunoki M., Sanjoh A. Structure-based design, synthesis, and evaluation of peptide-mimetic SARS 3CL protease inhibitors. Journal of Medicinal Chemistry. 2011;54(23):7962–7973. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

