Endoplasmic reticulum stress in amelogenesis imperfecta and phenotypic rescue using 4-phenylbutyrate
- PMID: 24362885
- PMCID: PMC3976337
- DOI: 10.1093/hmg/ddt642
Endoplasmic reticulum stress in amelogenesis imperfecta and phenotypic rescue using 4-phenylbutyrate
Abstract
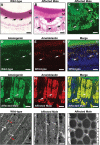
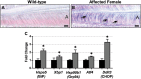
Inherited diseases caused by genetic mutations can arise due to loss of protein function. Alternatively, mutated proteins may mis-fold, impairing endoplasmic reticulum (ER) trafficking, causing ER stress and triggering the unfolded protein response (UPR). The UPR attempts to restore proteostasis but if unsuccessful drives affected cells towards apoptosis. Previously, we reported that in mice, the p.Tyr64His mutation in the enamel extracellular matrix (EEM) protein amelogenin disrupts the secretory pathway in the enamel-forming ameloblasts, resulting in eruption of malformed tooth enamel that phenocopies human amelogenesis imperfecta (AI). Defective amelogenin post-secretory self-assembly and processing within the developing EEM has been suggested to underlie the pathogenesis of X chromosome-linked AI. Here, we challenge this concept by showing that AI pathogenesis associated with the p.Tyr64His amelogenin mutation involves ameloblast apoptosis induced by ER stress. Furthermore, we show that 4-phenylbutyrate can rescue the enamel phenotype in affected female mice by promoting cell survival over apoptosis such that they are able to complete enamel formation despite the presence of the mutation, offering a potential therapeutic option for patients with this form of AI and emphasizing the importance of ER stress in the pathogenesis of this inherited conformational disease.
Figures




Similar articles
-
A mutation in the mouse Amelx tri-tyrosyl domain results in impaired secretion of amelogenin and phenocopies human X-linked amelogenesis imperfecta.Hum Mol Genet. 2010 Apr 1;19(7):1230-47. doi: 10.1093/hmg/ddq001. Epub 2010 Jan 12. Hum Mol Genet. 2010. PMID: 20067920 Free PMC article.
-
Amelogenesis imperfecta caused by N-terminal enamelin point mutations in mice and men is driven by endoplasmic reticulum stress.Hum Mol Genet. 2017 May 15;26(10):1863-1876. doi: 10.1093/hmg/ddx090. Hum Mol Genet. 2017. PMID: 28334996 Free PMC article.
-
AMELX Mutations and Genotype-Phenotype Correlation in X-Linked Amelogenesis Imperfecta.Int J Mol Sci. 2024 Jun 1;25(11):6132. doi: 10.3390/ijms25116132. Int J Mol Sci. 2024. PMID: 38892321 Free PMC article.
-
The Unfolded Protein Response in Amelogenesis and Enamel Pathologies.Front Physiol. 2017 Sep 8;8:653. doi: 10.3389/fphys.2017.00653. eCollection 2017. Front Physiol. 2017. PMID: 28951722 Free PMC article. Review.
-
Enamelin and autosomal-dominant amelogenesis imperfecta.Crit Rev Oral Biol Med. 2003;14(6):387-98. doi: 10.1177/154411130301400602. Crit Rev Oral Biol Med. 2003. PMID: 14656895 Review.
Cited by
-
Facilitation of Reparative Dentin Using a Drug Repositioning Approach With 4-Phenylbutric Acid.Front Physiol. 2022 May 4;13:885593. doi: 10.3389/fphys.2022.885593. eCollection 2022. Front Physiol. 2022. PMID: 35600310 Free PMC article.
-
Integrative Temporo-Spatial, Mineralogic, Spectroscopic, and Proteomic Analysis of Postnatal Enamel Development in Teeth with Limited Growth.Front Physiol. 2017 Oct 24;8:793. doi: 10.3389/fphys.2017.00793. eCollection 2017. Front Physiol. 2017. PMID: 29114228 Free PMC article.
-
Enamel Research: Priorities and Future Directions.Front Physiol. 2017 Jul 20;8:513. doi: 10.3389/fphys.2017.00513. eCollection 2017. Front Physiol. 2017. PMID: 28775693 Free PMC article. No abstract available.
-
Preparative SDS PAGE as an Alternative to His-Tag Purification of Recombinant Amelogenin.Front Physiol. 2017 Jun 16;8:424. doi: 10.3389/fphys.2017.00424. eCollection 2017. Front Physiol. 2017. PMID: 28670287 Free PMC article.
-
Gene cloning to clinical trials-the trials and tribulations of a life with collagen.Int J Exp Pathol. 2019 Feb;100(1):4-11. doi: 10.1111/iep.12311. Epub 2019 Mar 26. Int J Exp Pathol. 2019. PMID: 30912609 Free PMC article. Review.
References
-
- Hartl F.U., Bracher A., Hayer-Hartl M. Molecular chaperones in protein folding and proteostasis. Nature. 2011;475:324–332. doi:10.1038/nature10317. - DOI - PubMed
-
- Hartl F.U., Hayer-Hartl M. Converging concepts of protein folding in vitro and in vivo. Nat. Struct. Mol. Biol. 2009;16:574–581. doi:10.1038/nsmb.1591. - DOI - PubMed
-
- Ellisdon A.M., Bottomley S.P. The role of protein misfolding in the pathogenesis of human diseases. IUBMB Life. 2004;56:119–123. - PubMed
-
- Ozcan L., Tabas I. Role of endoplasmic reticulum stress in metabolic disease and other disorders. Annu. Rev. Med. 2012;63:317–328. doi:10.1146/annurev-med-043010-144749. - DOI - PMC - PubMed
-
- Yoshida H. ER stress and diseases. FEBS J. 2007;274:630–658. doi:10.1111/j.1742-4658.2007.05639.x. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

