Allosteric modulators hit the TSH receptor
- PMID: 24364583
- PMCID: PMC3868806
- DOI: 10.1210/en.2013-2079
Allosteric modulators hit the TSH receptor
Figures
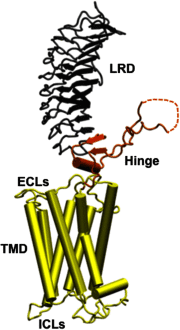
Comment on
-
A selective TSH receptor antagonist inhibits stimulation of thyroid function in female mice.Endocrinology. 2014 Jan;155(1):310-4. doi: 10.1210/en.2013-1835. Epub 2013 Dec 4. Endocrinology. 2014. PMID: 24169564 Free PMC article.
References
-
- Adams DD, Purves HD. Abnormal responses in the assay of thyrotropin. Proceedings of the University of Otago Medical School. 1956;34:11–12
-
- Smith BR, Hall R. Thyroid-stimulating immunoglobulins in Graves' disease. Lancet. 1974;2(7878):427–429 - PubMed
-
- Sanders J, Chirgadze DY, Sanders P, et al. Crystal structure of the TSH receptor in complex with a thyroid-stimulating autoantibody. Thyroid. 2007;17(5):395–410 - PubMed
-
- Calebiro D, Nikolaev VO, Persani L, Lohse MJ. Signaling by internalized G-protein-coupled receptors. Trends Pharmacol Sci. 2010;31(5):221–228 - PubMed
-
- Tanaka K, Chazenbalk GD, McLachlan SM, Rapoport B. Subunit structure of thyrotropin receptors expressed on the cell surface. J Biol Chem. 1999;274(48):33979–33984 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

