EBV infection is common in gingival epithelial cells of the periodontium and worsens during chronic periodontitis
- PMID: 24367478
- PMCID: PMC3868609
- DOI: 10.1371/journal.pone.0080336
EBV infection is common in gingival epithelial cells of the periodontium and worsens during chronic periodontitis
Abstract
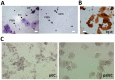
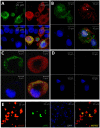
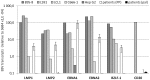
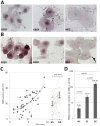
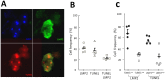
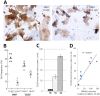
An amplifying role for oral epithelial cells (ECs) in Epstein-Barr Virus (EBV) infection has been postulated to explain oral viral shedding. However, while lytic or latent EBV infections of oro/nasopharyngeal ECs are commonly detected under pathological conditions, detection of EBV-infected ECs in healthy conditions is very rare. In this study, a simple non-surgical tissue sampling procedure was used to investigate EBV infection in the periodontal epithelium that surrounds and attaches teeth to the gingiva. Surprisingly, we observed that the gingival ECs of the periodontium (pECs) are commonly infected with EBV and may serve as an important oral reservoir of latently EBV-infected cells. We also found that the basal level of epithelial EBV-infection is significantly increased in chronic periodontitis, a common inflammatory disease that undermines the integrity of tooth-supporting tissues. Moreover, the level of EBV infection was found to correlate with disease severity. In inflamed tissues, EBV-infected pECs appear to be prone to apoptosis and to produce larger amounts of CCL20, a pivotal inflammatory chemokine that controls tissue infiltration by immune cells. Our discovery that the periodontal epithelium is a major site of latent EBV infection sheds a new light on EBV persistence in healthy carriers and on the role of this ubiquitous virus in periodontitis. Moreover, the identification of this easily accessible site of latent infection may encourage new approaches to investigate and monitor other EBV-associated disorders.
Conflict of interest statement
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

