A robust and quantitative method for tracking liposome contents after intravenous administration
- PMID: 24368300
- PMCID: PMC4040254
- DOI: 10.1016/j.jconrel.2013.12.014
A robust and quantitative method for tracking liposome contents after intravenous administration
Abstract
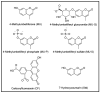
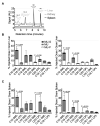
We introduce a method for tracking the rate and extent of delivery of liposome contents in vivo based on encapsulation of 4-methylumbelliferyl phosphate (MU-P), a profluorophore of 4-methylumbelliferone (MU). MU-P is rapidly dephosphorylated by endogenous phosphatases in vivo to form MU after leakage from the liposome. The change in fluorescence spectra when MU-P is converted to MU allows for quantification of entrapped (MU-P) and released (MU) liposome contents by fluorescence or by a sensitive high performance liquid chromatography assay. We define the "cellular availability" of an agent encapsulated in a liposome as the ratio of the amount of released agent in the tissue to the total amount of agent in the tissue; this parameter quantifies the fraction of drug available for therapy. The advantage of this method over existing technologies is the ability to decouple the signals of entrapped and released liposome contents. We validate this method by tracking the circulation and tissue distribution of MU-P loaded liposomes after intravenous administration. We use this assay to compare the cellular availability of liposomes composed of engineered phosphocholine lipids with covalently attached cholesterol, sterol-modified lipids (SML), to liposomes composed of conventional phospholipids and cholesterol. The SML liposomes have similar pharmacokinetic and biodistribution patterns as conventional phospholipid-cholesterol liposomes but a slower rate of contents delivery into the tissue. Thus, MU-P enables the tracking of the rate and extent of liposome contents release in tissues and should facilitate a better understanding of the pharmacodynamics of liposome-encapsulated drugs in animals.
Keywords: 4-methylumbelliferone; Drug release; Liposomes; Sterol-modified lipids.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures



References
-
- Zamboni WC, Gervais AC, Egorin MJ, Schellens JHM, Zuhowski EG, Pluim D, et al. Systemic and tumor disposition of platinum after administration of cisplatin or STEALTH liposomal-cisplatin formulations (SPI-077 and SPI-077 B103) in a preclinical tumor model of melanoma. Cancer Chemother. Pharmacol. 2004;53:329–336. - PubMed
-
- Drummond DC, Noble CO, Hayes ME, Park JW, Kirpotin DB. Pharmacokinetics and in vivo drug release rates in liposomal nanocarrier development. J Pharm Sci. 2008;97:4696–4740. - PubMed
-
- Thomas AM, Kapanen AI, Hare JI, Ramsay E, Edwards K, Karlsson G, et al. Development of a liposomal nanoparticle formulation of 5-fluorouracil for parenteral administration: formulation design, pharmacokinetics and efficacy. J Control Release. 2011;150:212–219. - PubMed
-
- Harrington KJ, Mohammadtaghi S, Uster PS, Glass D, Peters AM, Vile RG, et al. Effective targeting of solid tumors in patients with locally advanced cancers by radiolabeled pegylated liposomes. Clin Cancer Res. 2001;7:243–254. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

