GPR37 protein trafficking to the plasma membrane regulated by prosaposin and GM1 gangliosides promotes cell viability
- PMID: 24371137
- PMCID: PMC3931029
- DOI: 10.1074/jbc.M113.510883
GPR37 protein trafficking to the plasma membrane regulated by prosaposin and GM1 gangliosides promotes cell viability
Abstract
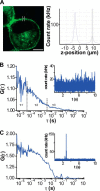
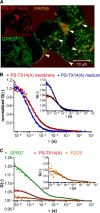
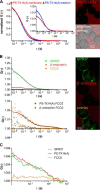
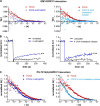
The subcellular distribution of the G protein-coupled receptor GPR37 affects cell viability and is implicated in the pathogenesis of parkinsonism. Intracellular accumulation and aggregation of GPR37 cause cell death, whereas GPR37 located in the plasma membrane provides cell protection. We define here a pathway through which the recently identified natural ligand, prosaposin, promotes plasma membrane association of GPR37. Immunoabsorption of extracellular prosaposin reduced GPR37(tGFP) surface density and decreased cell viability in catecholaminergic N2a cells. We found that GPR37(tGFP) partitioned in GM1 ganglioside-containing lipid rafts in the plasma membrane of live cells. This partitioning required extracellular prosaposin and was disrupted by lipid raft perturbation using methyl-β-cyclodextrin or cholesterol oxidase. Moreover, complex formation between GPR37(tGFP) and the GM1 marker cholera toxin was observed in the plasma membrane. These data show functional association between GPR37, prosaposin, and GM1 in the plasma membrane. These results thus tie together the three previously defined components of the cellular response to insult. Our findings identify a mechanism through which the receptor's natural ligand and GM1 may protect against toxic intracellular GPR37 aggregates observed in parkinsonism.
Keywords: Fluorescence Correlation Spectroscopy; G Protein-coupled Receptors (GPCR); Ganglioside; Lipid Raft; Pael-R; Parkinson Disease.
Figures




References
-
- Marazziti D., Gallo A., Golini E., Matteoni R., Tocchini-Valentini G. P. (1998) Molecular cloning and chromosomal localization of the mouse Gpr37 gene encoding an orphan G-protein-coupled peptide receptor expressed in brain and testis. Genomics 53, 315–324 - PubMed
-
- Zeng Z., Su K., Kyaw H., Li Y. (1997) A novel endothelin receptor type-B-like gene enriched in the brain. Biochem. Biophys. Res. Commun. 233, 559–567 - PubMed
-
- Imai Y., Soda M., Inoue H., Hattori N., Mizuno Y., Takahashi R. (2001) An unfolded putative transmembrane polypeptide, which can lead to endoplasmic reticulum stress, is a substrate of Parkin. Cell 105, 891–902 - PubMed
-
- Leng N., Gu G., Simerly R. B., Spindel E. R. (1999) Molecular cloning and characterization of two putative G protein-coupled receptors which are highly expressed in the central nervous system. Brain Res. Mol. Brain Res. 69, 73–83 - PubMed
-
- Murakami T., Shoji M., Imai Y., Inoue H., Kawarabayashi T., Matsubara E., Harigaya Y., Sasaki A., Takahashi R., Abe K. (2004) Pael-R is accumulated in Lewy bodies of Parkinson's disease. Ann. Neurol. 55, 439–442 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

