Functional limitations of plasmacytoid dendritic cells limit type I interferon, T cell responses and virus control in early life
- PMID: 24376875
- PMCID: PMC3871569
- DOI: 10.1371/journal.pone.0085302
Functional limitations of plasmacytoid dendritic cells limit type I interferon, T cell responses and virus control in early life
Abstract
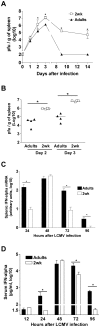
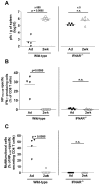
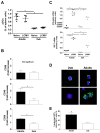
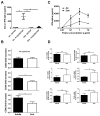
Infant mortality from viral infection remains a major global health concern: viruses causing acute infections in immunologically mature hosts often follow a more severe course in early life, with prolonged or persistent viral replication. Similarly, the WE strain of lymphocytic choriomeningitis virus (LCMV-WE) causes acute self-limiting infection in adult mice but follows a protracted course in infant animals, in which LCMV-specific CD8⁺ T cells fail to expand and control infection. By disrupting type I IFNs signaling in adult mice or providing IFN-α supplementation to infant mice, we show here that the impaired early life T cell responses and viral control result from limited early type I IFN responses. We postulated that plasmacytoid dendritic cells (pDC), which have been identified as one major source of immediate-early IFN-I, may not exert adult-like function in vivo in the early life microenvironment. We tested this hypothesis by studying pDC functions in vivo during LCMV infection and identified a coordinated downregulation of infant pDC maturation, activation and function: despite an adult-like in vitro activation capacity of infant pDCs, the expression of the E2-2 pDC master regulator (and of critical downstream antiviral genes such as MyD88, TLR7/TLR9, NF-κB, IRF7 and IRF8) is downregulated in vivo at baseline and during LCMV infection. A similar pattern was observed in response to ssRNA polyU, a model ligand of the TLR7 viral sensor. This suggests that the limited T cell-mediated defense against early life viral infections is largely attributable to / regulated by infant pDC responses and provides incentives for novel strategies to supplement or stimulate immediate-early IFN-α responses.
Conflict of interest statement
Figures

Similar articles
-
Siglec-H-Deficient Mice Show Enhanced Type I IFN Responses, but Do Not Develop Autoimmunity After Influenza or LCMV Infections.Front Immunol. 2021 Aug 23;12:698420. doi: 10.3389/fimmu.2021.698420. eCollection 2021. Front Immunol. 2021. PMID: 34497606 Free PMC article.
-
Hematopoietic and nonhematopoietic cells promote Type I interferon- and TLR7-dependent monocytosis during low-dose LCMV infection.Eur J Immunol. 2015 Nov;45(11):3064-72. doi: 10.1002/eji.201445331. Epub 2015 Sep 14. Eur J Immunol. 2015. PMID: 26289159 Free PMC article.
-
IRF9 Prevents CD8+ T Cell Exhaustion in an Extrinsic Manner during Acute Lymphocytic Choriomeningitis Virus Infection.J Virol. 2017 Oct 27;91(22):e01219-17. doi: 10.1128/JVI.01219-17. Print 2017 Nov 15. J Virol. 2017. PMID: 28878077 Free PMC article.
-
Complexities of Type I Interferon Biology: Lessons from LCMV.Viruses. 2019 Feb 20;11(2):172. doi: 10.3390/v11020172. Viruses. 2019. PMID: 30791575 Free PMC article. Review.
-
Viral escape mechanisms--escapology taught by viruses.Int J Exp Pathol. 2001 Oct;82(5):269-86. doi: 10.1046/j.1365-2613.2001.00204.x. Int J Exp Pathol. 2001. PMID: 11703537 Free PMC article. Review.
Cited by
-
Influence of Age and Immunostimulation on the Level of Toll-Like Receptor Gene (TLR3, 4, and 7) Expression in Foals.Animals (Basel). 2020 Oct 26;10(11):1966. doi: 10.3390/ani10111966. Animals (Basel). 2020. PMID: 33114637 Free PMC article.
-
Early life vaccination: Generation of adult-quality memory CD8+ T cells in infant mice using non-replicating adenoviral vectors.Sci Rep. 2016 Dec 8;6:38666. doi: 10.1038/srep38666. Sci Rep. 2016. PMID: 27929135 Free PMC article.
-
Ontogeny of myeloid cells.Front Immunol. 2014 Sep 3;5:423. doi: 10.3389/fimmu.2014.00423. eCollection 2014. Front Immunol. 2014. PMID: 25232355 Free PMC article. Review.
-
ETO family protein Mtg16 regulates the balance of dendritic cell subsets by repressing Id2.J Exp Med. 2014 Jul 28;211(8):1623-35. doi: 10.1084/jem.20132121. Epub 2014 Jun 30. J Exp Med. 2014. PMID: 24980046 Free PMC article.
-
Regulation of Transcription Factor E2-2 in Human Plasmacytoid Dendritic Cells by Monocyte-Derived TNFα.Viruses. 2020 Jan 31;12(2):162. doi: 10.3390/v12020162. Viruses. 2020. PMID: 32023836 Free PMC article.
References
-
- Ruest A, Michaud S, Deslandes S, Frost EH (2003) Comparison of the Directigen flu A+B test, the QuickVue influenza test, and clinical case definition to viral culture and reverse transcription-PCR for rapid diagnosis of influenza virus infection. J Clin Microbiol 41: 3487-3493. doi:10.1128/JCM.41.8.3487-3493.2003. PubMed: 12904343. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

