Binding Specificity of Philyra pisum Lectin to Pathogen-Associated Molecular Patterns, and Its Secondary Structure
- PMID: 24381505
- PMCID: PMC3874443
- DOI: 10.4196/kjpp.2013.17.6.547
Binding Specificity of Philyra pisum Lectin to Pathogen-Associated Molecular Patterns, and Its Secondary Structure
Abstract
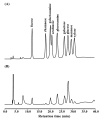
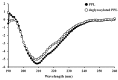
We recently reported a Philyra pisum lectin (PPL) that exerts mitogenic effects on human lymphocytes, and its molecular characterization. The present study provides a more detailed characterization of PPL based on the results from a monosaccharide analysis indicating that PPL is a glycoprotein, and circular dichroism spectra revealing its estimated α-helix, β-sheet, β-turn, and random coil contents to be 14.0%, 39.6%, 15.8%, and 30.6%, respectively. These contents are quite similar to those of deglycosylated PPL, indicating that glycans do not affect its intact structure. The binding properties to different pathogen-associated molecular patterns were investigated with hemagglutination inhibition assays using lipoteichoic acid from Gram-positive bacteria, lipopolysaccharide from Gram-negative bacteria, and both mannan and β-1,3-glucan from fungi. PPL binds to lipoteichoic acids and mannan, but not to lipopolysaccharides or β-1,3-glucan. PPL exerted no significant antiproliferative effects against human breast or bladder cancer cells. These results indicate that PPL is a glycoprotein with a lipoteichoic acid or mannan-binding specificity and which contains low and high proportions of α-helix and β-structures, respectively. These properties are inherent to the innate immune system of P. pisum and indicate that PPL could be involved in signal transmission into Gram-positive bacteria or fungi.
Keywords: Antiproliferative activity; Deglycosylation; Lectin; Pathogen-associated molecular patterns; Secondary structure.
Figures
Similar articles
-
Molecular characterization and mitogenic activity of a lectin from purse crab philyra pisum.Korean J Physiol Pharmacol. 2011 Aug;15(4):241-4. doi: 10.4196/kjpp.2011.15.4.241. Epub 2011 Aug 31. Korean J Physiol Pharmacol. 2011. PMID: 21994481 Free PMC article.
-
Isolation, characterization and molecular evolution of a novel pearl shell lectin from a marine bivalve, Pteria penguin.Mol Divers. 2006 Nov;10(4):607-18. doi: 10.1007/s11030-006-9051-3. Epub 2006 Nov 17. Mol Divers. 2006. PMID: 17111088
-
A Mesorhizobium lipopolysaccharide (LPS) specific lectin (CRL) from the roots of nodulating host plant, Cicer arietinum.Biochimie. 2011 Mar;93(3):440-9. doi: 10.1016/j.biochi.2010.10.017. Epub 2010 Nov 3. Biochimie. 2011. PMID: 21055439
-
[A turning point in the knowledge of the structure-function-activity relations of elastin].J Soc Biol. 2001;195(2):181-93. J Soc Biol. 2001. PMID: 11727705 Review. French.
-
Collectins and collectin receptors in innate immunity.APMIS Suppl. 2000;100:1-59. APMIS Suppl. 2000. PMID: 11021254 Review.
Cited by
-
Comparison and Optimization of Quantification Methods for Shigella flexneri Serotype 6 O-antigen Containing Galacturonic Acid and Methyl-Pentose.Int J Mol Sci. 2021 Nov 10;22(22):12160. doi: 10.3390/ijms222212160. Int J Mol Sci. 2021. PMID: 34830042 Free PMC article.
References
-
- Medzhitov R, Janeway CA., Jr Innate immunity: the virtues of a nonclonal system of recognition. Cell. 1997;91:295–298. - PubMed
-
- Lee SY, Söderhäll K. Early events in crustacean innate immunity. Fish Shellfish Immunol. 2002;12:421–437. - PubMed
-
- Medzhitov R, Janeway C., Jr Innate immune recognition: mechanisms and pathways. Immunol Rev. 2000;173:89–97. - PubMed
-
- Kurata S, Ariki S, Kawabata S. Recognition of pathogens and activation of immune responses in Drosophila and horseshoe crab innate immunity. Immunobiology. 2006;211:237–249. - PubMed
-
- Hoffmann JA, Kafatos FC, Janeway CA, Ezekowitz RA. Phylogenetic perspectives in innate immunity. Science. 1999;284:1313–1318. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources