Characterization of early pathogenesis in the SOD1(G93A) mouse model of ALS: part II, results and discussion
- PMID: 24381813
- PMCID: PMC3869683
- DOI: 10.1002/brb3.142
Characterization of early pathogenesis in the SOD1(G93A) mouse model of ALS: part II, results and discussion
Abstract
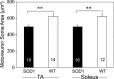
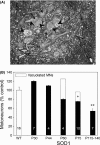
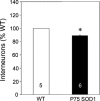
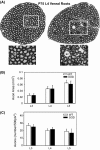
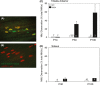
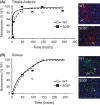
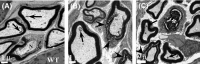
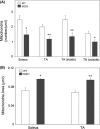
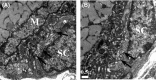
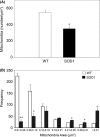
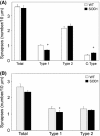
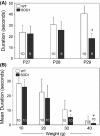
Pathological events are well characterized in amyotrophic lateral sclerosis (ALS) mouse models, but review of the literature fails to identify a specific initiating event that precipitates disease pathology. There is now growing consensus in the field that axon and synapses are first cellular sites of degeneration, but controversy exists over whether axon and synapse loss is initiated autonomously at those sites or by pathology in the cell body, in nonneuronal cells or even in nonmotoneurons (MNs). Previous studies have identified pathological events in the mutant superoxide dismutase 1 (SOD1) models involving spinal cord, peripheral axons, neuromuscular junctions (NMJs), or muscle; however, few studies have systematically examined pathogenesis at multiple sites in the same study. We have performed ultrastructural examination of both central and peripheral components of the neuromuscular system in the SOD1(G93A) mouse model of ALS. Twenty percent of MNs undergo degeneration by P60, but NMJ innervation in fast fatigable muscles is reduced by 40% by P30. Gait alterations and muscle weakness were also found at P30. There was no change in axonal transport prior to initial NMJ denervation. Mitochondrial morphological changes are observed at P7 and become more prominent with disease progression. At P30 there was a significant decrease in excitatory axo-dendritic and axo-somatic synapses with an increase in C-type axo-somatic synapses. Our study examined early pathology in both peripheral and central neuromuscular system. The muscle denervation is associated with functional motor deficits and begins during the first postnatal month in SOD1(G93A) mice. Physiological dysfunction and pathology in the mitochondria of synapses and MN soma and dendrites occur, and disease onset in these animals begins more than 2 months earlier than originally thought. This information may be valuable for designing preclinical trials that are more likely to impact disease onset and progression.
Keywords: Axons; NMJs; cytoplasmic vacuoles; glia; mega-mitochondria; mitochondria; motoneurons; motor function.
Figures













References
-
- Amendola J, Gueritaud JP, d'Incamps BL, Bories C, Liabeuf S, Allene C, et al. Postnatal electrical and morphological abnormalities in lumbar motoneurons from transgenic mouse models of amyotrophic lateral sclerosis. Arch. Ital. Biol. 2007;145:311–323. - PubMed
-
- Andrews JM, Gardner MB. Lower motor neuron degeneration associated with type C RNA virus infection in mice: neuropathological features. J. Neuropathol. Exp. Neurol. 1974;33:285–307. - PubMed
-
- Barbeito LH, Pehar M, Cassina P, Vargas MR, Peluffo H, Viera L, et al. A role for astrocytes in motor neuron loss in amyotrophic lateral sclerosis. Brain Res. Brain Res. Rev. 2004;47:263–274. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

