Heme oxygenase in neonatal lung injury and repair
- PMID: 24382006
- PMCID: PMC4203111
- DOI: 10.1089/ars.2013.5791
Heme oxygenase in neonatal lung injury and repair
Abstract
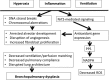
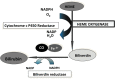
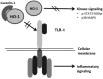
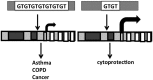
Significance: Premature and sick neonates are often exposed to high concentrations of oxygen, which results in lung injury and long-term adverse consequences. Nevertheless, neonates are more tolerant to hyperoxia than are adults. This may be, in part, explained by the high lung content of heme oxygenase-1 (HO-1), the rate-limiting enzyme in the degradation of heme and an important stress protein. The abundance of HO-1 dictates its cytoprotective and deleterious effects. Interestingly, in response to hyperoxia, lung HO-1 mRNA is not further up-regulated in neonates, suggesting that lung HO-1 gene expression is tightly regulated so as to optimize cytoprotection when faced with an oxidative stress such as hyperoxia.
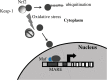
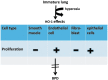
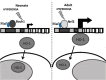
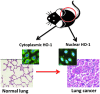
Recent advances: In addition to the lack of induction of HO-1 mRNA, neonatal lung HO-1 protein is observed in the nucleus in neonatal mice exposed to hyperoxia but not in adults, which is further evidence for the developmental regulation of HO-1. Nuclear HO-1 had unique properties independent of its enzymatic activity. In addition, there has been increasing evidence that nuclear HO-1 contributes to cellular proliferation and malignant transformation in several human cancers.
Critical issues: Since HO-1 has dual effects in cytoprotection and cellular proliferation, the titration of HO-1 effects is critical to ensure beneficial actions against oxidative stress.
Future directions: Much more has to be understood about the specific roles of HO-1 so as to manipulate its abundance and/or nuclear migration to maximize the therapeutic benefit of this pleiotropic protein in the neonatal lung.
Figures
Similar articles
-
Expression level and subcellular localization of heme oxygenase-1 modulates its cytoprotective properties in response to lung injury: a mouse model.PLoS One. 2014 Mar 5;9(3):e90936. doi: 10.1371/journal.pone.0090936. eCollection 2014. PLoS One. 2014. PMID: 24599172 Free PMC article.
-
Heme oxygenase-1 regulates postnatal lung repair after hyperoxia: role of β-catenin/hnRNPK signaling.Redox Biol. 2013 Feb 8;1(1):234-43. doi: 10.1016/j.redox.2013.01.013. eCollection 2013. Redox Biol. 2013. PMID: 24024157 Free PMC article.
-
Bach1 modulates heme oxygenase-1 expression in the neonatal mouse lung.Pediatr Res. 2009 Feb;65(2):145-9. doi: 10.1203/PDR.0b013e318191eedc. Pediatr Res. 2009. PMID: 18948842 Free PMC article.
-
Heme oxygenase-1: function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury.Am J Respir Cell Mol Biol. 1996 Jul;15(1):9-19. doi: 10.1165/ajrcmb.15.1.8679227. Am J Respir Cell Mol Biol. 1996. PMID: 8679227 Review.
-
A review on heme oxygenase-1 induction: is it a necessary evil.Inflamm Res. 2018 Jul;67(7):579-588. doi: 10.1007/s00011-018-1151-x. Epub 2018 Apr 24. Inflamm Res. 2018. PMID: 29693710 Review.
Cited by
-
Fucoidan attenuates hyperoxia-induced lung injury in newborn rats by mediating lung fibroblasts differentiate into myofibroblasts.Ann Transl Med. 2020 Nov;8(22):1501. doi: 10.21037/atm-20-6601. Ann Transl Med. 2020. PMID: 33313246 Free PMC article.
-
Heme Oxygenase-1 Supports Mitochondrial Energy Production and Electron Transport Chain Activity in Cultured Lung Epithelial Cells.Int J Mol Sci. 2020 Sep 22;21(18):6941. doi: 10.3390/ijms21186941. Int J Mol Sci. 2020. PMID: 32971746 Free PMC article.
-
Clinical Significance of Heme Oxygenase 1 in Tumor Progression.Antioxidants (Basel). 2021 May 17;10(5):789. doi: 10.3390/antiox10050789. Antioxidants (Basel). 2021. PMID: 34067625 Free PMC article. Review.
-
Two Faces of Heme Catabolic Pathway in Newborns: A Potential Role of Bilirubin and Carbon Monoxide in Neonatal Inflammatory Diseases.Oxid Med Cell Longev. 2020 Aug 18;2020:7140496. doi: 10.1155/2020/7140496. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32908636 Free PMC article. Review.
-
Neonatal lung function and therapeutics.Antioxid Redox Signal. 2014 Nov 1;21(13):1819-22. doi: 10.1089/ars.2014.5959. Epub 2014 Sep 16. Antioxid Redox Signal. 2014. PMID: 25111369 Free PMC article.
References
-
- Adamson IY, Young L, and Orr FW. Tumor metastasis after hyperoxic injury and repair of the pulmonary endothelium. Lab Invest 57: 71–77, 1987 - PubMed
-
- Alam J. and Cook JL. Transcriptional regulation of the heme oxygenase-1 gene via the stress response element pathway. Curr Pharm Des 9: 2499–2511, 2003 - PubMed
-
- Alam J, Stewart D, Touchard C, Boinapally S, Choi AM, and Cook JL. Nrf2, a Cap'n'Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J Biol Chem 274: 26071–26078, 1999 - PubMed
-
- An L, Liu CT, Qin XB, Liu QH, Liu Y, and Yu SY. Protective effects of hemin in an experimental model of ventilator-induced lung injury. Eur J Pharmacol 661: 102–108, 2011 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

