The candidate splicing factor Sfswap regulates growth and patterning of inner ear sensory organs
- PMID: 24391519
- PMCID: PMC3879212
- DOI: 10.1371/journal.pgen.1004055
The candidate splicing factor Sfswap regulates growth and patterning of inner ear sensory organs
Abstract
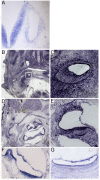
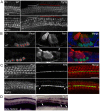
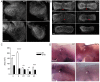
The Notch signaling pathway is thought to regulate multiple stages of inner ear development. Mutations in the Notch signaling pathway cause disruptions in the number and arrangement of hair cells and supporting cells in sensory regions of the ear. In this study we identify an insertional mutation in the mouse Sfswap gene, a putative splicing factor, that results in mice with vestibular and cochlear defects that are consistent with disrupted Notch signaling. Homozygous Sfswap mutants display hyperactivity and circling behavior consistent with vestibular defects, and significantly impaired hearing. The cochlea of newborn Sfswap mutant mice shows a significant reduction in outer hair cells and supporting cells and ectopic inner hair cells. This phenotype most closely resembles that seen in hypomorphic alleles of the Notch ligand Jagged1 (Jag1). We show that Jag1; Sfswap compound mutants have inner ear defects that are more severe than expected from simple additive effects of the single mutants, indicating a genetic interaction between Sfswap and Jag1. In addition, expression of genes involved in Notch signaling in the inner ear are reduced in Sfswap mutants. There is increased interest in how splicing affects inner ear development and function. Our work is one of the first studies to suggest that a putative splicing factor has specific effects on Notch signaling pathway members and inner ear development.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Lewis AK, Frantz GD, Carpenter DA, de Sauvage FJ, Gao WQ (1998) Distinct expression patterns of notch family receptors and ligands during development of the mammalian inner ear. Mech Dev 78: 159–163. - PubMed
-
- Murata J, Tokunaga A, Okano H, Kubo T (2006) Mapping of notch activation during cochlear development in mice: implications for determination of prosensory domain and cell fate diversification. J Comp Neurol 497: 502–518. - PubMed
-
- Morrison A, Hodgetts C, Gossler A, Hrabe de Angelis M, Lewis J (1999) Expression of Delta1 and Serrate1 (Jagged1) in the mouse inner ear. Mech Dev 84: 169–172. - PubMed
-
- Hartman BH, Hayashi T, Nelson BR, Bermingham-McDonogh O, Reh TA (2007) Dll3 is expressed in developing hair cells in the mammalian cochlea. Dev Dyn 236: 2875–2883. - PubMed
-
- Kiernan AE, Cordes R, Kopan R, Gossler A, Gridley T (2005) The Notch ligands DLL1 and JAG2 act synergistically to regulate hair cell development in the mammalian inner ear. Development 132: 4353–4362. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

