The role of syndecan-1 in cellular signaling and its effects on heparan sulfate biosynthesis in mesenchymal tumors
- PMID: 24392351
- PMCID: PMC3867677
- DOI: 10.3389/fonc.2013.00310
The role of syndecan-1 in cellular signaling and its effects on heparan sulfate biosynthesis in mesenchymal tumors
Abstract
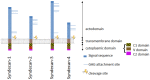
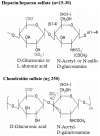
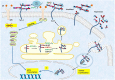
Proteoglycans (PGs) and in particular the syndecans are involved in the differentiation process across the epithelial-mesenchymal axis, principally through their ability to bind growth factors and modulate their downstream signaling. Malignant tumors have individual proteoglycan profiles, which are closely associated with their differentiation and biological behavior, mesenchymal tumors showing a different profile from that of epithelial tumors. Syndecan-1 is the main syndecan of epithelial malignancies, whereas in sarcomas its expression level is generally low, in accordance with their mesenchymal phenotype and highly malignant behavior. This proteoglycan is often overexpressed in adenocarcinoma cells, whereas mesothelioma and fibrosarcoma cells express syndecan-2 and syndecan-4 more abundantly. Increased expression of syndecan-1 in mesenchymal tumors changes the tumor cell morphology to an epithelioid direction whereas downregulation results in a change in shape from polygonal to spindle-like morphology. Although syndecan-1 plays major roles on the cell-surface, there are also intracellular functions, which are not very well studied. On the functional level, syndecan-1 affects mesenchymal tumor cell proliferation, adhesion, migration and motility, and the effect varies with the different domains of the core protein. Syndecan-1 may exert stimulatory or inhibitory effects, depending on the concentration of various mitogens, enzymes, and signaling molecules, the ratio between the shed and membrane-associated syndecan-1 and histological grade of the tumour. Growth factor signaling seems to be delicately controlled by regulatory loops involving the syndecan expression levels and their sulfation patterns. Overexpression of syndecan-1 modulates the biosynthesis and sulfation of heparan sulfate and it also affects the expression of other PGs. On transcriptomic level, syndecan-1 modulation results in profound effects on genes involved in regulation of cell growth.
Keywords: cancer; heparan sulfate; mesenchymal tumor; signaling; syndecan-1.
Figures
References
-
- Marynen P, Zhang J, Cassiman JJ, Van den Berghe H, David G. Partial primary structure of the 48- and 90-kilodalton core proteins of cell surface-associated heparan sulfate proteoglycans of lung fibroblasts. Prediction of an integral membrane domain and evidence for multiple distinct core proteins at the cell surface of human lung fibroblasts. J Biol Chem (1989) 264(12):7017–24 - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

