The oxidation states of DJ-1 dictate the cell fate in response to oxidative stress triggered by 4-hpr: autophagy or apoptosis?
- PMID: 24392637
- PMCID: PMC4158984
- DOI: 10.1089/ars.2013.5446
The oxidation states of DJ-1 dictate the cell fate in response to oxidative stress triggered by 4-hpr: autophagy or apoptosis?
Abstract
Aim: Chemotherapy-induced reactive oxygen species (ROS) not only contribute to apoptosis, but also trigger autophagy. Since autophagy is reported to protect cancer cells from apoptosis, this weakens the therapeutic effect of chemotherapy. This study aimed at identifying the key molecules that determine the cellular response to ROS and, therefore, provide better strategies to increase chemotherapeutic efficiency.
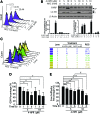
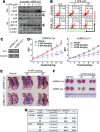
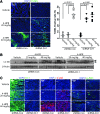
Results: Increasing concentrations of N-(4-hydroxyphenyl) retinamide (4-HPR)-treatment pushed autophagy down to apoptosis in a dose-dependent manner, and 4-HPR-induced ROS contribute to this process. Since we found that ASK1-regulated JNK1 and p38 are responsible for 4-HPR-induced autophagy and apoptosis, respectively, we further utilized co-immunoprecipitation followed by liquid chromatography-tandem mass spectrometry analysis to identify proteins that specifically bind to ASK1 under different oxidative states. Of note, DJ-1, a crucial antioxidant protein, was identified. Interestingly, DJ-1 functions as a redox sensor that senses ROS levels and determines the cellular response to 4-HPR: Under mild oxidative stress, moderate oxidation of DJ-1 is recruited to inhibit the activity of ASK1 and maintain cell viability by activating autophagy; under a lethal level of oxidative stress, excessive oxidized DJ-1 dissociates from ASK1 and activates it, thereby initiating p38 activation and enabling the cells to commit to apoptosis. Moreover, the depletion of DJ-1 increases the sensitivity of tumor cells to 4-HPR both in vitro and in vivo.
Innovation: Our results reveal that the different oxidation states of DJ-1 function as a cellular redox sensor of ROS caused by 4-HPR and determine the cell fate of autophagy or apoptosis. Moreover, the results suggest that DJ-1 might be a potent therapeutic target for cancer treatment.
Conclusion: ROS-mediated changes in the oxidation state of DJ-1 are involved in 4-HPR's effect on pushing autophagy down to apoptosis. Consequently, this change mediates ASK1 activation by regulating DJ-1-ASK1 complex formation and determines the cell fate of autophagy or apoptosis.
Figures





References
-
- Boulares AH, Yakovlev AG, Ivanova V, Stoica BA, Wang G, Iyer S, and Smulson M. Role of poly(ADP-ribose) polymerase (PARP) cleavage in apoptosis. Caspase 3-resistant PARP mutant increases rates of apoptosis in transfected cells. J Biol Chem 274: 22932–22940, 1999 - PubMed
-
- Byun JY, Yoon CH, An S, Park IC, Kang CM, Kim MJ, and Lee SJ. The Rac1/MKK7/JNK pathway signals upregulation of Atg5 and subsequent autophagic cell death in response to oncogenic Ras. Carcinogenesis 30: 1880–1888, 2009 - PubMed
-
- Cai YJ, Lu JJ, Zhu H, Xie H, Huang M, Lin LP, Zhang XW, and Ding J. Salvicine triggers DNA double-strand breaks and apoptosis by GSH-depletion-driven H2O2 generation and topoisomerase II inhibition. Free Radic Biol Med 45: 627–635, 2008 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

