Intrinsic properties of and thalamocortical inputs onto identified corticothalamic-VPM neurons
- PMID: 24397568
- PMCID: PMC6995656
- DOI: 10.3109/08990220.2013.869495
Intrinsic properties of and thalamocortical inputs onto identified corticothalamic-VPM neurons
Abstract
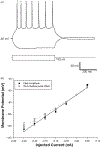
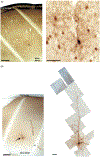
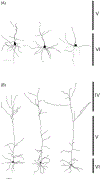
Corticothalamic (CT) feedback plays an important role in regulating the sensory information that the cortex receives. Within the somatosensory cortex layer VI originates the feedback to the ventral posterior medial (VPM) nucleus of the thalamus, which in turn receives sensory information from the contralateral whiskers. We examined the physiology and morphology of CT neurons in rat somatosensory cortex, focusing on the physiological characteristics of the monosynaptic inputs that they receive from the thalamus. To identify CT neurons, rhodamine microspheres were injected into VPM and allowed to retrogradely transport to the soma of CT neurons. Thalamocortical slices were prepared at least 3 days post injection. Whole-cell recordings from labeled CT cells in layer VI demonstrated that they are regular spiking neurons and exhibit little spike frequency adaption. Two anatomical classes were identified based on their apical dendrites that either terminated by layer V (compact cells) or layer IV (elaborate cells). Thalamic inputs onto identified CT-VPM neurons demonstrated paired pulse depression over a wide frequency range (2-20 Hz). Stimulus trains also resulted in significant synaptic depression above 10 Hz. Our results suggest that thalamic inputs differentially impact CT-VPM neurons in layer VI. This characteristic may allow them to differentiate a wide range of stimulation frequencies which in turn further tune the feedback signals to the thalamus.
Keywords: Barrel cortex; layer VI; neuronal morphology.
Conflict of interest statement
Declaration of interest
The mention of commercial products, their sources, or their use in connection with material reported herein is not to be construed as either an actual or implied endorsement of such products by the Department of Health and Human Services. The authors have no further disclosures.
Figures

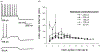


Similar articles
-
VPM and PoM nuclei of the rat somatosensory thalamus: intrinsic neuronal properties and corticothalamic feedback.Cereb Cortex. 2007 Dec;17(12):2853-65. doi: 10.1093/cercor/bhm025. Epub 2007 Mar 26. Cereb Cortex. 2007. PMID: 17389627
-
Cortex dynamically modulates responses of thalamic relay neurons through prolonged circuit-level disinhibition in rat thalamus in vivo.J Neurophysiol. 2016 Nov 1;116(5):2368-2382. doi: 10.1152/jn.00424.2016. Epub 2016 Aug 31. J Neurophysiol. 2016. PMID: 27582292 Free PMC article.
-
The excitatory neuronal network of rat layer 4 barrel cortex.J Neurosci. 2000 Oct 15;20(20):7579-86. doi: 10.1523/JNEUROSCI.20-20-07579.2000. J Neurosci. 2000. PMID: 11027217 Free PMC article.
-
Corticothalamic Pathways in Auditory Processing: Recent Advances and Insights From Other Sensory Systems.Front Neural Circuits. 2021 Aug 19;15:721186. doi: 10.3389/fncir.2021.721186. eCollection 2021. Front Neural Circuits. 2021. PMID: 34489648 Free PMC article. Review.
-
Dynamics of sensory thalamocortical synaptic networks during information processing states.Prog Neurobiol. 2004 Nov;74(4):213-47. doi: 10.1016/j.pneurobio.2004.09.002. Prog Neurobiol. 2004. PMID: 15556288 Review.
Cited by
-
A corticothalamic switch: controlling the thalamus with dynamic synapses.Neuron. 2015 May 6;86(3):768-82. doi: 10.1016/j.neuron.2015.03.040. Epub 2015 Apr 23. Neuron. 2015. PMID: 25913856 Free PMC article.
-
Exercises in Anatomy, Connectivity, and Morphology using Neuromorpho.org and the Allen Brain Atlas.J Undergrad Neurosci Educ. 2015 Mar 15;13(2):A95-A100. eCollection 2015 Spring. J Undergrad Neurosci Educ. 2015. PMID: 25838808 Free PMC article.
-
Muscarinic and Nicotinic Modulation of Neocortical Layer 6A Synaptic Microcircuits Is Cooperative and Cell-Specific.Cereb Cortex. 2020 May 18;30(6):3528-3542. doi: 10.1093/cercor/bhz324. Cereb Cortex. 2020. PMID: 32026946 Free PMC article.
-
Corticothalamic modulation of somatosensory thalamic tactile processing.J Physiol. 2025 May;603(9):2801-2819. doi: 10.1113/JP287526. Epub 2025 Apr 3. J Physiol. 2025. PMID: 40178512 Free PMC article.
-
Thalamocortical and Intracortical Inputs Differentiate Layer-Specific Mouse Auditory Corticocollicular Neurons.J Neurosci. 2019 Jan 9;39(2):256-270. doi: 10.1523/JNEUROSCI.3352-17.2018. Epub 2018 Oct 25. J Neurosci. 2019. PMID: 30361396 Free PMC article.
References
-
- Agmon A, Connors BW. 1991. Thalamocortical responses of mouse somatosensory (barrel) cortex in vitro. Neuroscience 41:365–379. - PubMed
-
- Ahissar E, Sosnik R, Haidarliu S. 2000. Transformation from temporal to rate coding in a somatosensory thalamocortical pathway. Nature 406: 302–306. - PubMed
-
- Beierlein M, Connors BW. 2002. Short-term dynamics of thalamocortical and intracortical synapses onto layer 6 neurons in neocortex. J Neurophysiol 88:1924–1932. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
