Pro-inflammatory role of natural killer cells in the development of allergic airway disease
- PMID: 24397722
- PMCID: PMC3985123
- DOI: 10.1111/cea.12271
Pro-inflammatory role of natural killer cells in the development of allergic airway disease
Abstract
Background: Natural Killer (NK) cells have been implicated in the development of allergic airway inflammation. However, the in vivo role of NK cells has not been firmly established due to the lack of animal models with selective deficiencies in NK cells.
Objective: To determine the specific contribution of NK cells in a murine model of allergic airway disease (AAD).
Methods: The role of NK cells in AAD was studied using NK-deficient (NKD) mice, perforin(-/-) mice, and mice depleted of Ly49A/D/G(+) NK cell subsets in an ovalbumin-induced model of allergic airway disease (OVA-AAD).
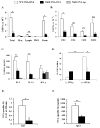
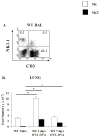
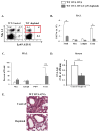
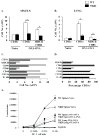
Results: Induction of OVA-AAD in C57BL/6 wild-type (WT) mice resulted in the expansion of airway NK cells and the development of pronounced airway eosinophilia. In the absence of NK cells or specific subsets of NK cells, either in NKD mice, or after the depletion of Ly49A/D/G(+) NK cells, the development of OVA-AAD was significantly impaired as seen by decreased airway inflammation and eosinophilia, decreased secretion of the Th2 cytokines IL-4, IL-5 and IL-13 and diminished OVA-specific antibody production. Furthermore, while OVA-exposure induced a dramatic expansion of dendritic cells (DCs) in WT mice, their induction was significantly attenuated in NKD mice. Development of OVA-AAD in perforin(-/-) mice suggested that the proinflammatory role of NK cells is not dependent on perforin-mediated cytotoxicity. Lastly, induction of allergic disease by OVA-specific CD4 T cells from WT but not NK-depleted or NKD mice in RAG(-/-) recipients, demonstrates that NK cells are essential for T cell priming.
Conclusions and clinical relevance: Our data demonstrate that conventional NK cells play an important and distinct role in the development of AAD. The presence of activated NK cells has been noted in patients with asthma. Understanding the mechanisms by which NK cells regulate allergic disease is therefore an important component of treatment approaches.
Keywords: allergic airway disease; allergic inflammation; asthma; natural killer T cells; natural killer cells.
© 2014 John Wiley & Sons Ltd.
Figures



Similar articles
-
Natural Killer Cells in the Lungs.Front Immunol. 2019 Jun 25;10:1416. doi: 10.3389/fimmu.2019.01416. eCollection 2019. Front Immunol. 2019. PMID: 31293580 Free PMC article. Review.
-
IL-15-deficient mice develop enhanced allergic responses to airway allergen exposure.Clin Exp Allergy. 2017 May;47(5):639-655. doi: 10.1111/cea.12886. Epub 2017 Feb 7. Clin Exp Allergy. 2017. PMID: 28093832 Free PMC article.
-
Plasticity of invariant NKT cell regulation of allergic airway disease is dependent on IFN-gamma production.J Immunol. 2010 Jul 1;185(1):253-62. doi: 10.4049/jimmunol.0902301. Epub 2010 Jun 4. J Immunol. 2010. PMID: 20525882
-
Cutting edge: invariant V alpha 14 NKT cells are required for allergen-induced airway inflammation and hyperreactivity in an experimental asthma model.J Immunol. 2003 Aug 15;171(4):1637-41. doi: 10.4049/jimmunol.171.4.1637. J Immunol. 2003. PMID: 12902459
-
Emerging insights into human health and NK cell biology from the study of NK cell deficiencies.Immunol Rev. 2019 Jan;287(1):202-225. doi: 10.1111/imr.12725. Immunol Rev. 2019. PMID: 30565241 Free PMC article. Review.
Cited by
-
Maternal diesel particle exposure promotes offspring asthma through NK cell-derived granzyme B.J Clin Invest. 2020 Aug 3;130(8):4133-4151. doi: 10.1172/JCI130324. J Clin Invest. 2020. PMID: 32407293 Free PMC article.
-
Maintenance of Type 2 Response by CXCR6-Deficient ILC2 in Papain-Induced Lung Inflammation.Int J Mol Sci. 2019 Nov 4;20(21):5493. doi: 10.3390/ijms20215493. Int J Mol Sci. 2019. PMID: 31690060 Free PMC article.
-
Allergen-induced activation of natural killer cells represents an early-life immune response in the development of allergic asthma.J Allergy Clin Immunol. 2018 Dec;142(6):1856-1866. doi: 10.1016/j.jaci.2018.02.019. Epub 2018 Mar 5. J Allergy Clin Immunol. 2018. PMID: 29518416 Free PMC article.
-
Natural Killer Cells in the Lungs.Front Immunol. 2019 Jun 25;10:1416. doi: 10.3389/fimmu.2019.01416. eCollection 2019. Front Immunol. 2019. PMID: 31293580 Free PMC article. Review.
-
Curcumin Ingestion Inhibits Mastocytosis and Suppresses Intestinal Anaphylaxis in a Murine Model of Food Allergy.PLoS One. 2015 Jul 6;10(7):e0132467. doi: 10.1371/journal.pone.0132467. eCollection 2015. PLoS One. 2015. PMID: 26147007 Free PMC article.
References
-
- Azzawi M, Bradley B, Jeffery PK, Frew AJ, Wardlaw AJ, Knowles G, Assoufi B, Collins JV, Durham S, Kay AB. Identification of activated T lymphocytes and eosinophils in bronchial biopsies in stable atopic asthma. Am Rev Respir Dis. 1990;142:1407–13. - PubMed
-
- Lukacs NW, Strieter RM, Chensue SW, Kunkel SL. Interleukin-4-dependent pulmonary eosinophil infiltration in a murine model of asthma. Am J Respir Cell Mol Biol. 1994;10:526–32. - PubMed
-
- Robinson DS, Hamid Q, Ying S, Tsicopoulos A, Barkans J, Bentley AM, Corrigan C, Durham SR, Kay AB. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med. 1992;326:298–304. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

