High-resolution imaging of cardiomyocyte behavior reveals two distinct steps in ventricular trabeculation
- PMID: 24401373
- PMCID: PMC3899815
- DOI: 10.1242/dev.098632
High-resolution imaging of cardiomyocyte behavior reveals two distinct steps in ventricular trabeculation
Abstract
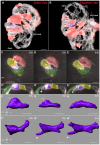
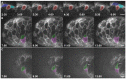
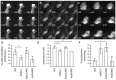
Over the course of development, the vertebrate heart undergoes a series of complex morphogenetic processes that transforms it from a simple myocardial epithelium to the complex 3D structure required for its function. One of these processes leads to the formation of trabeculae to optimize the internal structure of the ventricle for efficient conduction and contraction. Despite the important role of trabeculae in the development and physiology of the heart, little is known about their mechanism of formation. Using 3D time-lapse imaging of beating zebrafish hearts, we observed that the initiation of cardiac trabeculation can be divided into two processes. Before any myocardial cell bodies have entered the trabecular layer, cardiomyocytes extend protrusions that invade luminally along neighboring cell-cell junctions. These protrusions can interact within the trabecular layer to form new cell-cell contacts. Subsequently, cardiomyocytes constrict their abluminal surface, moving their cell bodies into the trabecular layer while elaborating more protrusions. We also examined the formation of these protrusions in trabeculation-deficient animals, including erbb2 mutants, tnnt2a morphants, which lack cardiac contractions and flow, and myh6 morphants, which lack atrial contraction and exhibit reduced flow. We found that, compared with cardiomyocytes in wild-type hearts, those in erbb2 mutants were less likely to form protrusions, those in tnnt2a morphants formed less stable protrusions, and those in myh6 morphants extended fewer protrusions per cell. Thus, through detailed 4D imaging of beating hearts, we have identified novel cellular behaviors underlying cardiac trabeculation.
Keywords: Cardiac cell biology; Live imaging; Morphogenesis; Trabeculation; Zebrafish; erbb2.
Figures


References
-
- Beis D., Stainier D. Y. R. (2006). In vivo cell biology: following the zebrafish trend. Trends Cell Biol. 16, 105–112 - PubMed
-
- Berdougo E., Coleman H., Lee D. H., Stainier D. Y. R., Yelon D. (2003). Mutation of weak atrium/atrial myosin heavy chain disrupts atrial function and influences ventricular morphogenesis in zebrafish. Development 130, 6121–6129 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

