Neuroprotective effects of donepezil against cholinergic depletion
- PMID: 24401551
- PMCID: PMC3978431
- DOI: 10.1186/alzrt215
Neuroprotective effects of donepezil against cholinergic depletion
Abstract
Introduction: Intraparenchymal injections of the immunotoxin 192-IgG-saporin into medial septum and nucleus basalis magnocellularis causes a selective depletion of basal forebrain cholinergic neurons. Thus, it represents a valid model to mimic a key component of the cognitive deficits associated with aging and dementia. Here we administered donepezil, a potent acetylcholinesterase inhibitor developed for treating Alzheimer's disease, 15 days before 192-IgG-saporin injection, and thus we examined donepezil effects on neurodegeneration and cognitive deficits.
Methods: Caspase-3 activity and cognitive performances of lesioned rats pre-treated with donepezil or saline were analyzed and compared to the outcomes obtained in pre-treated sham-lesioned rats.
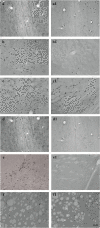
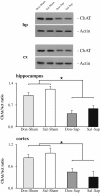
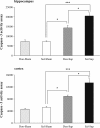
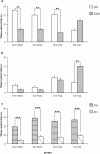
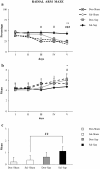
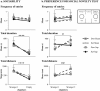
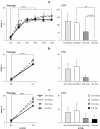
Results: Cholinergic depletion increased hippocampal and neocortical caspase-3 activity and impaired working memory, spatial discrimination, social novelty preference, and ultrasonic vocalizations, without affecting anxiety levels and fear conditioning. In lesioned animals, donepezil pre-treatment reduced hippocampal and neocortical caspase-3 activity and improved working memory and spatial discrimination performances and partially rescued ultrasonic vocalizations, without preventing social novelty alterations.
Conclusions: Present data indicate that donepezil pre-treatment exerts beneficial effects on behavioral deficits induced by cholinergic depletion, attenuating the concomitant hippocampal and neocortical neurodegeneration.
Figures

Similar articles
-
Chronic Lithium Treatment in a Rat Model of Basal Forebrain Cholinergic Depletion: Effects on Memory Impairment and Neurodegeneration.J Alzheimers Dis. 2017;56(4):1505-1518. doi: 10.3233/JAD-160892. J Alzheimers Dis. 2017. PMID: 28222508
-
Cognitive performances of cholinergically depleted rats following chronic donepezil administration.J Alzheimers Dis. 2009;17(1):161-76. doi: 10.3233/JAD-2009-1040. J Alzheimers Dis. 2009. PMID: 19221411
-
Intraparenchymal infusions of 192 IgG-saporin: development of a method for selective and discrete lesioning of cholinergic basal forebrain nuclei.J Neurosci Methods. 1999 Sep 15;91(1-2):9-19. doi: 10.1016/s0165-0270(99)00057-6. J Neurosci Methods. 1999. PMID: 10522820
-
Immunolesion by 192IgG-saporin of rat basal forebrain cholinergic system: a useful tool to produce cortical cholinergic dysfunction.Prog Brain Res. 1996;109:253-64. doi: 10.1016/s0079-6123(08)62109-3. Prog Brain Res. 1996. PMID: 9009714 Review.
-
[Donepezil-induced neuroprotection of acetylcholinergic neurons in olfactory bulbectomized mice].Yakugaku Zasshi. 2010 May;130(5):717-21. doi: 10.1248/yakushi.130.717. Yakugaku Zasshi. 2010. PMID: 20460870 Review. Japanese.
Cited by
-
Fluoxetine and Ketamine Reverse the Depressive but Not Anxiety Behavior Induced by Lesion of Cholinergic Neurons in the Horizontal Limb of the Diagonal Band of Broca in Male Rat.Front Behav Neurosci. 2021 Feb 18;15:602708. doi: 10.3389/fnbeh.2021.602708. eCollection 2021. Front Behav Neurosci. 2021. PMID: 33679340 Free PMC article.
-
Changes in Neuroimmune and Neuronal Death Markers after Adolescent Alcohol Exposure in Rats are Reversed by Donepezil.Sci Rep. 2019 Aug 20;9(1):12110. doi: 10.1038/s41598-019-47039-1. Sci Rep. 2019. PMID: 31431637 Free PMC article.
-
Donepezil Reverses Dendritic Spine Morphology Adaptations and Fmr1 Epigenetic Modifications in Hippocampus of Adult Rats After Adolescent Alcohol Exposure.Alcohol Clin Exp Res. 2018 Apr;42(4):706-717. doi: 10.1111/acer.13599. Epub 2018 Feb 15. Alcohol Clin Exp Res. 2018. PMID: 29336496 Free PMC article.
-
Streptozotocin Intracerebroventricular-Induced Neurotoxicity and Brain Insulin Resistance: a Therapeutic Intervention for Treatment of Sporadic Alzheimer's Disease (sAD)-Like Pathology.Mol Neurobiol. 2016 Sep;53(7):4548-62. doi: 10.1007/s12035-015-9384-y. Epub 2015 Aug 23. Mol Neurobiol. 2016. PMID: 26298663 Review.
-
Effects of Omega-3 Fatty Acid Supplementation on Cognitive Functions and Neural Substrates: A Voxel-Based Morphometry Study in Aged Mice.Front Aging Neurosci. 2016 Mar 4;8:38. doi: 10.3389/fnagi.2016.00038. eCollection 2016. Front Aging Neurosci. 2016. PMID: 26973513 Free PMC article.
References
-
- Bartus RT, Dean RL 3rd, Beer B, Lippa AS. The cholinergic hypothesis of geriatric memory dysfunction. Science. 1982;5:408–414. - PubMed
-
- Bartus RT. On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Exp Neurol. 2000;5:495–529. - PubMed
-
- Sarter M, Bruno JP. Developmental origins of the age-related decline in cortical cholinergic function and associated cognitive abilities. Neurobiol Aging. 2004;5:1127–1139. - PubMed
-
- Galimberti D, Scarpini E. Treatment of Alzheimer’s disease: symptomatic and disease-modifying approaches. Curr Aging Sci. 2010;5:46–56. - PubMed
-
- Schwarz S, Froelich L, Burns A. Pharmacological treatment of dementia. Curr Opin Psychiatry. 2012;5:542–550. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

