Diverse thalamocortical short-term plasticity elicited by ongoing stimulation
- PMID: 24403151
- PMCID: PMC6608150
- DOI: 10.1523/JNEUROSCI.2441-13.2014
Diverse thalamocortical short-term plasticity elicited by ongoing stimulation
Abstract
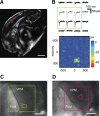
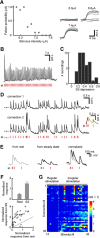
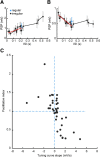
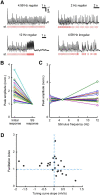
To produce sensation, neuronal pathways must transmit and process stimulus patterns that unfold over time. This behavior is determined by short-term synaptic plasticity (STP), which shapes the temporal filtering properties of synapses in a pathway. We explored STP variability across thalamocortical (TC) synapses, measuring whole-cell responses to stimulation of TC fibers in layer 4 neurons of mouse barrel cortex in vitro. As expected, STP during stimulation from rest was dominated by depression. However, STP during ongoing stimulation was strikingly diverse across TC connections. Diversity took the form of variable tuning to the latest interstimulus interval: some connections responded weakly to shorter intervals, while other connections were facilitated. These behaviors did not cluster into categories but formed a continuum. Diverse tuning did not require disynaptic inhibition. Hence, monosynaptic excitatory lemniscal TC connections onto layer 4 do not behave uniformly during ongoing stimulation. Each connection responds differentially to particular stimulation intervals, enriching the ability of the pathway to convey complex, temporally fluctuating information.
Figures

References
-
- Abbott LF, Varela JA, Sen K, Nelson SB. Synaptic depression and cortical gain control. Science. 1997;275:220–224. - PubMed
-
- Bale MR, Ince RAA, Santagata G, Petersen RS. Population coding of texture-induced whisker motion in the ventroposterior medial nucleus of the thalamus. Soc Neurosci Abstr. 2011;37:704.14.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
