Hypothalamic dysfunction of the thrombospondin receptor α2δ-1 underlies the overeating and obesity triggered by brain-derived neurotrophic factor deficiency
- PMID: 24403154
- PMCID: PMC3870936
- DOI: 10.1523/JNEUROSCI.1572-13.2014
Hypothalamic dysfunction of the thrombospondin receptor α2δ-1 underlies the overeating and obesity triggered by brain-derived neurotrophic factor deficiency
Abstract
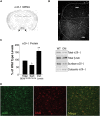
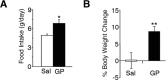
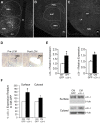
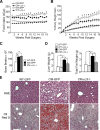
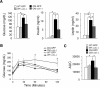
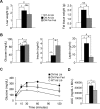
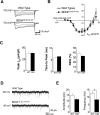
Brain-derived neurotrophic factor (BDNF) and its receptor, TrkB, are critical components of the neural circuitry controlling appetite and body weight. Diminished BDNF signaling in mice results in severe hyperphagia and obesity. In humans, BDNF haploinsufficiency and the functional Bdnf Val66Met polymorphism have been linked to elevated food intake and body weight. The mechanisms underlying this dysfunction are poorly defined. We demonstrate a chief role of α2δ-1, a calcium channel subunit and thrombospondin receptor, in triggering overeating in mice with central BDNF depletion. We show reduced α2δ-1 cell-surface expression in the BDNF mutant ventromedial hypothalamus (VMH), an energy balance-regulating center. This deficit contributes to the hyperphagia exhibited by BDNF mutant mice because selective inhibition of α2δ-1 by gabapentin infusion into wild-type VMH significantly increases feeding and body weight gain. Importantly, viral-mediated α2δ-1 rescue in BDNF mutant VMH significantly mitigates their hyperphagia, obesity, and liver steatosis and normalizes deficits in glucose homeostasis. Whole-cell recordings in BDNF mutant VMH neurons revealed normal calcium currents but reduced frequency of EPSCs. These results suggest calcium channel-independent effects of α2δ-1 on feeding and implicate α2δ-1-thrombospondin interactions known to facilitate excitatory synapse assembly. Our findings identify a central mechanism mediating the inhibitory effects of BDNF on feeding. They also demonstrate a novel and critical role for α2δ-1 in appetite control and suggest a mechanism underlying weight gain in humans treated with gabapentinoid drugs.
Figures
Comment in
-
Neuroscience study uncovers new player in obesity.Am J Manag Care. 2014 Jan;20(1 Spec No.):E11. Am J Manag Care. 2014. PMID: 25928938 No abstract available.
Similar articles
-
Sex-specific Effects of α2δ-1 in the Ventromedial Hypothalamus of Female Mice Controlling Glucose and Lipid Balance.Endocrinology. 2020 Jul 1;161(7):bqaa068. doi: 10.1210/endocr/bqaa068. Endocrinology. 2020. PMID: 32337532 Free PMC article.
-
Selective deletion of Bdnf in the ventromedial and dorsomedial hypothalamus of adult mice results in hyperphagic behavior and obesity.J Neurosci. 2007 Dec 26;27(52):14265-74. doi: 10.1523/JNEUROSCI.3308-07.2007. J Neurosci. 2007. PMID: 18160634 Free PMC article.
-
Regulation of Satiety by Bdnf-e2-Expressing Neurons through TrkB Activation in Ventromedial Hypothalamus.Biomolecules. 2023 May 11;13(5):822. doi: 10.3390/biom13050822. Biomolecules. 2023. PMID: 37238691 Free PMC article.
-
The rise, fall, and resurrection of the ventromedial hypothalamus in the regulation of feeding behavior and body weight.Physiol Behav. 2006 Feb 28;87(2):221-44. doi: 10.1016/j.physbeh.2005.10.007. Epub 2006 Jan 18. Physiol Behav. 2006. PMID: 16412483 Review.
-
A putative model of overeating and obesity based on brain-derived neurotrophic factor: direct and indirect effects.Behav Neurosci. 2012 Aug;126(4):505-14. doi: 10.1037/a0028600. Epub 2012 Jun 11. Behav Neurosci. 2012. PMID: 22687148 Review.
Cited by
-
Sex-specific Effects of α2δ-1 in the Ventromedial Hypothalamus of Female Mice Controlling Glucose and Lipid Balance.Endocrinology. 2020 Jul 1;161(7):bqaa068. doi: 10.1210/endocr/bqaa068. Endocrinology. 2020. PMID: 32337532 Free PMC article.
-
The Role of BDNF and TrkB in the Central Control of Energy and Glucose Balance: An Update.Biomolecules. 2024 Mar 31;14(4):424. doi: 10.3390/biom14040424. Biomolecules. 2024. PMID: 38672441 Free PMC article. Review.
-
Auxiliary α2δ1 and α2δ3 Subunits of Calcium Channels Drive Excitatory and Inhibitory Neuronal Network Development.J Neurosci. 2020 Jun 17;40(25):4824-4841. doi: 10.1523/JNEUROSCI.1707-19.2020. Epub 2020 May 15. J Neurosci. 2020. PMID: 32414783 Free PMC article.
-
Alpha2delta-1 in SF1+ Neurons of the Ventromedial Hypothalamus Is an Essential Regulator of Glucose and Lipid Homeostasis.Cell Rep. 2017 Dec 5;21(10):2737-2747. doi: 10.1016/j.celrep.2017.11.048. Cell Rep. 2017. PMID: 29212022 Free PMC article.
-
Ventromedial hypothalamic expression of Bdnf is required to establish normal patterns of afferent GABAergic connectivity and responses to hypoglycemia.Mol Metab. 2015 Dec 15;5(2):91-101. doi: 10.1016/j.molmet.2015.11.007. eCollection 2016 Feb. Mol Metab. 2015. PMID: 26909317 Free PMC article.
References
-
- Aguado F, Carmona MA, Pozas E, Aguiló A, Martínez-Guijarro FJ, Alcantara S, Borrell V, Yuste R, Ibañez CF, Soriano E. BDNF regulates spontaneous correlated activity at early developmental stages by increasing synaptogenesis and expression of the K+/Cl− co-transporter KCC2. Development. 2003;130:1267–1280. doi: 10.1242/dev.00351. - DOI - PubMed
-
- Bennett J, Duan D, Engelhardt JF, Maguire AM. Real-time, noninvasive in vivo assessment of adeno-associated virus-mediated retinal transduction. Invest Ophthalmol Vis Sci. 1997;38:2857–2863. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
