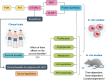
Therapeutic potential and adverse events of everolimus for treatment of hepatocellular carcinoma - systematic review and meta-analysis
- PMID: 24403259
- PMCID: PMC3892390
- DOI: 10.1002/cam4.150
Therapeutic potential and adverse events of everolimus for treatment of hepatocellular carcinoma - systematic review and meta-analysis
Abstract
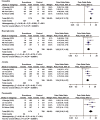
Everolimus is an orally administrated mammalian target of rapamycin (mTOR) inhibitor. Several large-scale randomized controlled trials (RCTs) have demonstrated the survival benefits of everolimus at the dose of 10 mg/day for solid cancers. Furthermore, mTOR-inhibitor-based immunosuppression is associated with survival benefits for patients with hepatocellular carcinoma (HCC) who have received liver transplantation. However, a low rate of tumor reduction and some adverse events have been pointed out. This review summarizes the antitumor effects and adverse events of everolimus and evaluates its possible application in advanced HCC. For the meta-analysis of adverse events, we used the RCTs for solid cancers. The odds ratios of adverse events were calculated using the Peto method. Manypreclinical studies demonstrated that everolimus had antitumor effects such as antiproliferation and antiangiogenesis. However, some differences in the effects were observed among in vivo animal studies for HCC treatment. Meanwhile, clinical studies demonstrated that the response rate of single-agent everolimus was low, though survival benefits could be expected. The meta-analysis revealed the odds ratios (95% confidence interval [CI]) of stomatitis: 5.42 [4.31-6.73], hyperglycemia: 3.22 [2.37-4.39], anemia: 3.34 [2.37-4.67], pneumonitis: 6.02 [3.95-9.16], aspartate aminotransferase levels: 2.22 [1.37-3.62], and serum alanine aminotransferase levels: 2.94 [1.72-5.02], respectively. Everolimus at the dose of 10 mg/day significantly increased the risk of the adverse events. In order to enable its application to the standard conventional therapies of HCC, further studies are required to enhance the antitumor effects and manage the adverse events of everolimus.
Keywords: Adverse events; everolimus; hepatocellular carcinoma; liver transplantation.
© 2013 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Figures
References
-
- Llovet JM, Bruix J. Novel advancements in the management of hepatocellular carcinoma in 2008. J. Hepatol. 2008;48:12. - PubMed
-
- Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008;359:378–390. - PubMed
-
- Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous