Stimulated bronchial epithelial cells release bioactive lysophosphatidylcholine 16:0, 18:0, and 18:1
- PMID: 24404396
- PMCID: PMC3881404
- DOI: 10.4168/aair.2014.6.1.66
Stimulated bronchial epithelial cells release bioactive lysophosphatidylcholine 16:0, 18:0, and 18:1
Abstract
Purpose: In human subjects and animal models with acute and chronic lung injury, the bioactive lysophosphatidylcholine (LPC) is elevated in lung lining fluids. The increased LPC can promote an inflammatory microenvironment resulting in lung injury. Furthermore, pathological lung conditions are associated with upregulated phospholipase A2 (PLA2), the predominant enzyme producing LPC in tissues by hydrolysis of phosphatidylcholine. However, the lung cell populations responsible for increases of LPC have yet to be systematically characterized. The goal was to investigate the LPC generation by bronchial epithelial cells in response to pathological mediators and determine the major LPC species produced.
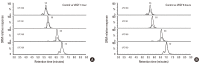
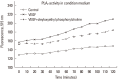
Methods: Primary human bronchial epithelial cells (NHBE) were challenged by vascular endothelial growth factor (VEGF) for 1 or 6 h, and condition medium and cells collected for quantification of predominant LPC species by high performance liquid chromatography-tandem mass spectrometry (LC-MS-MS). The cells were analyzed by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) for PLA2. The direct effects of LPC in inducing inflammatory activities on NHBE were assessed by transepithelial resistance as well as expression of interleukin-8 (IL-8) and matrix metalloproteinase-1 (MMP-1).
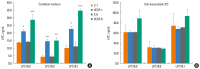
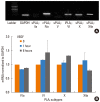
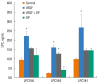
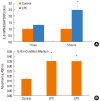
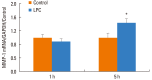
Results: VEGF stimulation of NHBE for 1 or 6 h, significantly increased concentrations of LPC16:0, LPC18:0, and LPC18:1 in condition medium compared to control. The sPLA2-selective inhibitor (oleyloxyethyl phosphorylcholine) inhibited the VEGF-induced release of LPC16:0 and LPC18:1 and PLA2 activity. In contrast, NHBE stimulated with TNF did not induce LPC release. VEGF did not increase mRNA of PLA2 subtypes sPLA2-X, sPLA2-XIIa, cPLA2-IVa, and iPLA2-VI. Exogenous LPC treatment increased expression of IL-8 and MMP-1, and reduced the transepithelial resistance in NHBE.
Conclusions: Our findings indicate that VEGF-stimulated bronchial epithelial cells are a key source of extracellular LPCs, which can function as an autocrine mediator with potential to induce airway epithelial inflammatory injury.
Keywords: Bronchial epithelium; lysophosphatidylcholine; phospholipase A2.
Conflict of interest statement
There are no financial or other issues that might lead to conflict of interest.
Figures

References
-
- Nakos G, Kitsiouli EI, Tsangaris I, Lekka ME. Bronchoalveolar lavage fluid characteristics of early intermediate and late phases of ARDS. Alterations in leukocytes, proteins, PAF and surfactant components. Intensive Care Med. 1998;24:296–303. - PubMed
-
- Arbibe L, Koumanov K, Vial D, Rougeot C, Faure G, Havet N, Longacre S, Vargaftig BB, Béréziat G, Voelker DR, Wolf C, Touqui L. Generation of lyso-phospholipids from surfactant in acute lung injury is mediated by type-II phospholipase A2 and inhibited by a direct surfactant protein A-phospholipase A2 protein interaction. J Clin Invest. 1998;102:1152–1160. - PMC - PubMed
-
- Rice KL, Duane PG, Archer SL, Gilboe DP, Niewoehner DE. H2O2 injury causes Ca(2+)-dependent and -independent hydrolysis of phosphatidylcholine in alveolar epithelial cells. Am J Physiol. 1992;263:L430–L438. - PubMed
-
- Schaefer CA, Kuhlmann CR, Gast C, Weiterer S, Li F, Most AK, Neumann T, Backenkohler U, Tillmanns H, Waldecker B, Wiecha J, Erdogan A. Statins prevent oxidized low-density lipoprotein- and lysophosphatidylcholine-induced proliferation of human endothelial cells. Vascul Pharmacol. 2004;41:67–73. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

