The neurodegenerative effects of selenium are inhibited by FOXO and PINK1/PTEN regulation of insulin/insulin-like growth factor signaling in Caenorhabditis elegans
- PMID: 24406377
- PMCID: PMC3979119
- DOI: 10.1016/j.neuro.2013.12.012
The neurodegenerative effects of selenium are inhibited by FOXO and PINK1/PTEN regulation of insulin/insulin-like growth factor signaling in Caenorhabditis elegans
Abstract
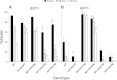
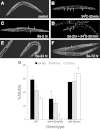
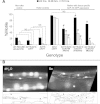
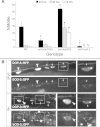
Exposures to high levels of environmental selenium have been associated with motor neuron disease in both animals and humans and high levels of selenite have been identified in the cerebrospinal fluid of patients with amyotrophic lateral sclerosis (ALS). We have shown previously that exposures to high levels of sodium selenite in the environment of Caenorhabditis elegans adult animals can induce neurodegeneration and cell loss resulting in motor deficits and death and that this is at least partially caused by a reduction in cholinergic signaling across the neuromuscular junction. Here we provide evidence that reduction in insulin/insulin-like (IIS) signaling alters response to high dose levels of environmental selenium which in turn can regulate the IIS pathway. Most specifically we show that nuclear localization and thus activation of the DAF-16/forkhead box transcription factor occurs in response to selenium exposure although this was not observed in motor neurons of the ventral cord. Yet, tissue specific expression and generalized overexpression of DAF-16 can partially rescue the neurodegenerative and behavioral deficits observed with high dose selenium exposures in not only the cholinergic, but also the GABAergic motor neurons. In addition, two modifiers of IIS signaling, PTEN (phosphatase and tensin homolog, deleted on chromosome 10) and PINK1 (PTEN-induced putative kinase 1) are required for the cellular antioxidant reduced glutathione to mitigate the selenium-induced movement deficits. Studies have suggested that environmental exposures can lead to ALS or other neurological diseases and this model of selenium-induced neurodegeneration developed in a genetically tractable organism provides a tool for examining the combined roles of genetics and environment in the neuro-pathologic disease process.
Keywords: Amyotrophic lateral sclerosis; Insulin/insulin-like signaling; Neurodegeneration; PINK1; PTEN; Selenium.
Copyright © 2014 The Authors. Published by Elsevier B.V. All rights reserved.
Figures



Similar articles
-
Selenium induces cholinergic motor neuron degeneration in Caenorhabditis elegans.Neurotoxicology. 2012 Oct;33(5):1021-32. doi: 10.1016/j.neuro.2012.04.019. Epub 2012 Apr 25. Neurotoxicology. 2012. PMID: 22560997 Free PMC article.
-
Role of Caenorhabditis elegans AKT-1/2 and SGK-1 in Manganese Toxicity.Neurotox Res. 2018 Oct;34(3):584-596. doi: 10.1007/s12640-018-9915-1. Epub 2018 Jun 7. Neurotox Res. 2018. PMID: 29882004 Free PMC article.
-
The C. elegans adult neuronal IIS/FOXO transcriptome reveals adult phenotype regulators.Nature. 2016 Jan 7;529(7584):92-6. doi: 10.1038/nature16483. Epub 2015 Dec 14. Nature. 2016. PMID: 26675724 Free PMC article.
-
DAF-16: FOXO in the Context of C. elegans.Curr Top Dev Biol. 2018;127:1-21. doi: 10.1016/bs.ctdb.2017.11.007. Epub 2018 Feb 2. Curr Top Dev Biol. 2018. PMID: 29433733 Review.
-
DAF-16/FoxO in Caenorhabditis elegans and Its Role in Metabolic Remodeling.Cells. 2020 Jan 2;9(1):109. doi: 10.3390/cells9010109. Cells. 2020. PMID: 31906434 Free PMC article. Review.
Cited by
-
FoxO proteins in the nervous system.Anal Cell Pathol (Amst). 2015;2015:569392. doi: 10.1155/2015/569392. Epub 2015 Jun 10. Anal Cell Pathol (Amst). 2015. PMID: 26171319 Free PMC article. Review.
-
GABAergic system's Injuries Induced by Sodium Sulfite in Caenorhabditis elegans Were Prevented by the Anti-Oxidative Properties of Dehydroepiandrosterone Sulfate.Neurotox Res. 2020 Aug;38(2):447-460. doi: 10.1007/s12640-020-00207-y. Epub 2020 May 14. Neurotox Res. 2020. PMID: 32410195
-
Role of dehydroepiandrosterone sulfate as a scavenging antioxidant: the evidence from Caenorhabditis elegans' nervous system under hypoxia.Neural Regen Res. 2021 Aug;16(8):1537-1538. doi: 10.4103/1673-5374.303022. Neural Regen Res. 2021. PMID: 33433471 Free PMC article. No abstract available.
-
Regeneration in the nervous system with erythropoietin.Front Biosci (Landmark Ed). 2016 Jan;21(3):561-596. doi: 10.2741/4408. Front Biosci (Landmark Ed). 2016. PMID: 26549969 Free PMC article.
-
Neuropharmaceutical Properties of Naringin Against Alzheimer's and Parkinson's Diseases: Naringin Protection Against AD and PD.Galen Med J. 2022 Nov 12;11:e2337. doi: 10.31661/gmj.v11i.2337. eCollection 2022. Galen Med J. 2022. PMID: 36698693 Free PMC article.
References
-
- Altun Z.F., Hall D.H. WormAtlas. 2011. Nervous system, general description.
-
- Andreassen O.A., Ferrante R.J., Klivenyi P., Klein A.M., Shinobu L.A., Epstein C.J. Partial deficiency of manganese superoxide dismutase exacerbates a transgenic mouse model of amyotrophic lateral sclerosis. Ann Neurol. 2000;47(4):447–455. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

