Delayed administration of a single dose of lithium promotes recovery from AKI
- PMID: 24408869
- PMCID: PMC3935583
- DOI: 10.1681/ASN.2013040350
Delayed administration of a single dose of lithium promotes recovery from AKI
Abstract
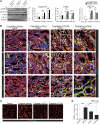
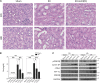
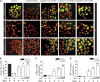
Evidence suggests that glycogen synthase kinase 3β (GSK3β) contributes to AKI; however, its role in post-AKI kidney repair remains uncertain. Here, delayed treatment with a single dose of lithium, a selective inhibitor of GSK3β and a US Food and Drug Administration-approved mood stabilizer, accelerated recovery of renal function, promoted repopulation of renal tubular epithelia, and improved kidney repair in murine models of cisplatin- and ischemia/reperfusion-induced AKI. These effects associated with reduced GSK3β activity and elevated expression of proproliferative molecules, including cyclin D1, c-Myc, and hypoxia-inducible factor 1α (HIF-1α), in renal tubular epithelia. In cultured renal tubular cells, cisplatin exposure led to transient repression of GSK3β activity followed by a prolonged upregulation of activity. Rescue treatment with lithium inhibited GSK3β activity, enhanced nuclear expression of cyclin D1, c-Myc, and HIF-1α, and boosted cellular proliferation. Similarly, ectopic expression of a kinase-dead mutant of GSK3β enhanced the expression of cyclin D1, c-Myc, and HIF-1α and amplified cellular proliferation after cisplatin injury, whereas forced expression of a constitutively active mutant of GSK3β abrogated the effects of lithium. Mechanistically, GSK3β colocalized and physically interacted with cyclin D1, c-Myc, and HIF-1α in tubular cells. In silico analysis revealed that cyclin D1, c-Myc, and HIF-1α harbor putative GSK3β consensus phosphorylation motifs, implying GSK3β-directed phosphorylation and subsequent degradation of these molecules. Notably, cotreatment with lithium enhanced the proapoptotic effects of cisplatin in cultured colon cancer cells. Collectively, our findings suggest that pharmacologic targeting of GSK3β by lithium may be a novel therapeutic strategy to improve renal salvage after AKI.
Figures




Comment in
-
Lithium in kidney diseases: big roles for the smallest metal.J Am Soc Nephrol. 2014 Mar;25(3):421-3. doi: 10.1681/ASN.2013111216. Epub 2014 Jan 9. J Am Soc Nephrol. 2014. PMID: 24408870 Free PMC article. No abstract available.
-
Basic research: the long and the short of it-the temporal effects of renal lithium exposure are beginning to be unravelled.Nat Rev Nephrol. 2014 Mar;10(3):123. doi: 10.1038/nrneph.2014.10. Epub 2014 Jan 28. Nat Rev Nephrol. 2014. PMID: 24468767 No abstract available.
References
-
- Humphreys BD, Valerius MT, Kobayashi A, Mugford JW, Soeung S, Duffield JS, McMahon AP, Bonventre JV: Intrinsic epithelial cells repair the kidney after injury. Cell Stem Cell 2: 284–291, 2008 - PubMed
-
- Witzgall R, Brown D, Schwarz C, Bonventre JV: Localization of proliferating cell nuclear antigen, vimentin, c-Fos, and clusterin in the postischemic kidney. Evidence for a heterogenous genetic response among nephron segments, and a large pool of mitotically active and dedifferentiated cells. J Clin Invest 93: 2175–2188, 1994 - PMC - PubMed
-
- Yang L, Humphreys BD, Bonventre JV: Pathophysiology of acute kidney injury to chronic kidney disease: Maladaptive repair. Contrib Nephrol 174: 149–155, 2011 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

