Unraveling the role of podocyte turnover in glomerular aging and injury
- PMID: 24408871
- PMCID: PMC3968496
- DOI: 10.1681/ASN.2013050452
Unraveling the role of podocyte turnover in glomerular aging and injury
Abstract
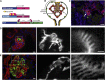
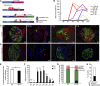
Podocyte loss is a major determinant of progressive CKD. Although recent studies showed that a subset of parietal epithelial cells can serve as podocyte progenitors, the role of podocyte turnover and regeneration in repair, aging, and nephron loss remains unclear. Here, we combined genetic fate mapping with highly efficient podocyte isolation protocols to precisely quantify podocyte turnover and regeneration. We demonstrate that parietal epithelial cells can give rise to fully differentiated visceral epithelial cells indistinguishable from resident podocytes and that limited podocyte renewal occurs in a diphtheria toxin model of acute podocyte ablation. In contrast, the compensatory programs initiated in response to nephron loss evoke glomerular hypertrophy, but not de novo podocyte generation. In addition, no turnover of podocytes could be detected in aging mice under physiologic conditions. In the absence of podocyte replacement, characteristic features of aging mouse kidneys included progressive accumulation of oxidized proteins, deposits of protein aggregates, loss of podocytes, and glomerulosclerosis. In summary, quantitative investigation of podocyte regeneration in vivo provides novel insights into the mechanism and capacity of podocyte turnover and regeneration in mice. Our data reveal that podocyte generation is mainly confined to glomerular development and may occur after acute glomerular injury, but it fails to regenerate podocytes in aging kidneys or in response to nephron loss.
Figures



References
-
- Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, McCullough PA, Kasiske BL, Kelepouris E, Klag MJ, Parfrey P, Pfeffer M, Raij L, Spinosa DJ, Wilson PW, American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention : Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Circulation 108: 2154–2169, 2003 - PubMed
-
- Zoja C, Abbate M, Remuzzi G: Progression of chronic kidney disease: I from animal models. Curr Opin Nephrol Hypertens 15: 250–257, 2006 - PubMed
-
- Wharram BL, Goyal M, Wiggins JE, Sanden SK, Hussain S, Filipiak WE, Saunders TL, Dysko RC, Kohno K, Holzman LB, Wiggins RC: Podocyte depletion causes glomerulosclerosis: Diphtheria toxin-induced podocyte depletion in rats expressing human diphtheria toxin receptor transgene. J Am Soc Nephrol 16: 2941–2952, 2005 - PubMed
-
- Wiggins JE, Goyal M, Sanden SK, Wharram BL, Shedden KA, Misek DE, Kuick RD, Wiggins RC: Podocyte hypertrophy, “adaptation,” and “decompensation” associated with glomerular enlargement and glomerulosclerosis in the aging rat: Prevention by calorie restriction. J Am Soc Nephrol 16: 2953–2966, 2005 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

