Sex differences in stress-induced social withdrawal: role of brain derived neurotrophic factor in the bed nucleus of the stria terminalis
- PMID: 24409132
- PMCID: PMC3885825
- DOI: 10.3389/fnbeh.2013.00223
Sex differences in stress-induced social withdrawal: role of brain derived neurotrophic factor in the bed nucleus of the stria terminalis
Abstract
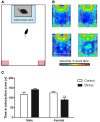
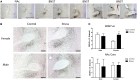
Depression and anxiety disorders are more common in women than men, and little is known about the neurobiological mechanisms that contribute to this disparity. Recent data suggest that stress-induced changes in neurotrophins have opposing effects on behavior by acting in different brain networks. Social defeat has been an important approach for understanding neurotrophin action, but low female aggression levels in rats and mice have limited the application of these methods primarily to males. We examined the effects of social defeat in monogamous California mice (Peromyscus californicus), a species in which both males and females defend territories. We demonstrate that defeat stress increases mature brain-derived neurotrophic factor (BDNF) protein but not mRNA in the bed nucleus of the stria terminalis (BNST) in females but not males. Changes in BDNF protein were limited to anterior subregions of the BNST, and there were no changes in the adjacent nucleus accumbens (NAc). The effects of defeat on social withdrawal behavior and BDNF were reversed by chronic, low doses of the antidepressant sertraline. However, higher doses of sertraline restored social withdrawal and elevated BDNF levels. Acute treatment with a low dose of sertraline failed to reverse the effects of defeat. Infusions of the selective tyrosine-related kinase B receptor (TrkB) antagonist ANA-12 into the anterior BNST specifically increased social interaction in stressed females but had no effect on behavior in females naïve to defeat. These results suggest that stress-induced increases in BDNF in the anterior BNST contribute to the exaggerated social withdrawal phenotype observed in females.
Keywords: BDNF; BNST; SSRI; dose; sex; social defeat.
Figures



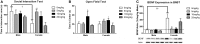

Similar articles
-
Sex differences in effects of dopamine D1 receptors on social withdrawal.Neuropharmacology. 2014 Feb;77:208-16. doi: 10.1016/j.neuropharm.2013.09.026. Epub 2013 Oct 10. Neuropharmacology. 2014. PMID: 24120838 Free PMC article.
-
Sex-specific effects of social defeat stress on miRNA expression in the anterior BNST.Behav Brain Res. 2021 Mar 5;401:113084. doi: 10.1016/j.bbr.2020.113084. Epub 2020 Dec 22. Behav Brain Res. 2021. PMID: 33358922 Free PMC article.
-
Oxytocin Receptors in the Anteromedial Bed Nucleus of the Stria Terminalis Promote Stress-Induced Social Avoidance in Female California Mice.Biol Psychiatry. 2018 Feb 1;83(3):203-213. doi: 10.1016/j.biopsych.2017.08.024. Epub 2017 Sep 14. Biol Psychiatry. 2018. PMID: 29066224 Free PMC article.
-
Sex differences in the effects of social defeat on brain and behavior in the California mouse: Insights from a monogamous rodent.Semin Cell Dev Biol. 2017 Jan;61:92-98. doi: 10.1016/j.semcdb.2016.06.021. Epub 2016 Jun 30. Semin Cell Dev Biol. 2017. PMID: 27375045 Free PMC article. Review.
-
Chemoarchitecture of the bed nucleus of the stria terminalis: Neurophenotypic diversity and function.Handb Clin Neurol. 2021;179:385-402. doi: 10.1016/B978-0-12-819975-6.00025-X. Handb Clin Neurol. 2021. PMID: 34225977 Free PMC article. Review.
Cited by
-
Inhibition of vasopressin V1a receptors in the medioventral bed nucleus of the stria terminalis has sex- and context-specific anxiogenic effects.Neuropharmacology. 2016 Nov;110(Pt A):59-68. doi: 10.1016/j.neuropharm.2016.07.018. Epub 2016 Jul 21. Neuropharmacology. 2016. PMID: 27452721 Free PMC article.
-
Quantifying Sex Differences in Behavior in the Era of "Big" Data.Cold Spring Harb Perspect Biol. 2022 May 27;14(5):a039164. doi: 10.1101/cshperspect.a039164. Cold Spring Harb Perspect Biol. 2022. PMID: 34607831 Free PMC article. Review.
-
Sex-dependent effects of developmental exposure to bisphenol A and ethinyl estradiol on metabolic parameters and voluntary physical activity.J Dev Orig Health Dis. 2015 Dec;6(6):539-52. doi: 10.1017/S2040174415001488. Epub 2015 Sep 18. J Dev Orig Health Dis. 2015. PMID: 26378919 Free PMC article.
-
Toward the explainability, transparency, and universality of machine learning for behavioral classification in neuroscience.Curr Opin Neurobiol. 2022 Apr;73:102544. doi: 10.1016/j.conb.2022.102544. Epub 2022 Apr 26. Curr Opin Neurobiol. 2022. PMID: 35487088 Free PMC article. Review.
-
Comparing behavior following binge ethanol in adolescent and adult DBA/2 J mice.Behav Brain Res. 2022 Feb 15;419:113703. doi: 10.1016/j.bbr.2021.113703. Epub 2021 Dec 3. Behav Brain Res. 2022. PMID: 34864163 Free PMC article.
References
-
- Babb J. A., Masini C. V., Day H. E. W., Campeau S. (2013). Sex differences in activated corticotropin-releasing factor neurons within stress-related neurocircuitry and hypothalamic–pituitary–adrenocortical axis hormones following restraint in rats. Neuroscience 234, 40–52 10.1016/j.neuroscience.2012.12.051 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

