CD13 promotes mesenchymal stem cell-mediated regeneration of ischemic muscle
- PMID: 24409152
- PMCID: PMC3885827
- DOI: 10.3389/fphys.2013.00402
CD13 promotes mesenchymal stem cell-mediated regeneration of ischemic muscle
Abstract
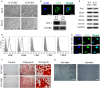
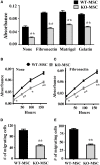
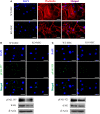
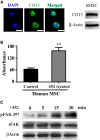
Mesenchymal stem cells (MSCs) are multipotent, tissue-resident cells that can facilitate tissue regeneration and thus, show great promise as potential therapeutic agents. Functional MSCs have been isolated and characterized from a wide array of adult tissues and are universally identified by the shared expression of a core panel of MSCs markers. One of these markers is the multifunctional cell surface peptidase CD13 that has been shown to be expressed on human and murine MSCs from many tissues. To investigate whether this universal expression indicates a functional role for CD13 in MSC biology we isolated, expanded and characterized MSCs from bone marrow of wild type (WT) and CD13(KO) mice. Characterization of these cells demonstrated that both WT and CD13(KO) MSCs expressed the full complement of MSC markers (CD29, CD44, CD49e, CD105, Sca1), showed comparable proliferation rates and were capable of differentiating toward the adipogenic and osteogenic lineages. However, MSCs lacking CD13 were unable to differentiate into vascular cells, consistent with our previous characterization of CD13 as an angiogenic regulator. Compared to WT MSCs, adhesion and migration on various extracellular matrices of CD13(KO) MSCs were significantly impaired, which correlated with decreased phospho-FAK levels and cytoskeletal alterations. Crosslinking human MSCs with activating CD13 antibodies increased cell adhesion to endothelial monolayers and induced FAK activation in a time dependent manner. In agreement with these in vitro data, intramuscular injection of CD13(KO) MSCs in a model of severe ischemic limb injury resulted in significantly poorer perfusion, decreased ambulation, increased necrosis and impaired vascularization compared to those receiving WT MSCs. This study suggests that CD13 regulates FAK activation to promote MSC adhesion and migration, thus, contributing to MSC-mediated tissue repair. CD13 may present a viable target to enhance the efficacy of mesenchymal stem cell therapies.
Keywords: CD13; adhesion; cell transplantation; hindlimb ischemia; mesenchymal stem cells.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

