Inhibition of HIF prolyl-4-hydroxylases by FG-4497 reduces brain tissue injury and edema formation during ischemic stroke
- PMID: 24409307
- PMCID: PMC3883663
- DOI: 10.1371/journal.pone.0084767
Inhibition of HIF prolyl-4-hydroxylases by FG-4497 reduces brain tissue injury and edema formation during ischemic stroke
Abstract
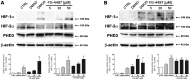
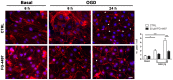
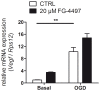
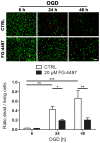
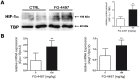
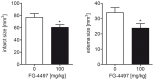
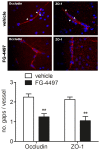
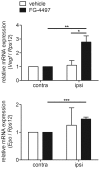
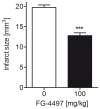
Ischemic stroke results in disruption of the blood-brain barrier (BBB), edema formation and neuronal cell loss. Some neuroprotective factors such as vascular endothelial growth factor (VEGF) favor edema formation, while others such as erythropoietin (Epo) can mitigate it. Both factors are controlled by hypoxia inducible transcription factors (HIF) and the activity of prolyl hydroxylase domain proteins (PHD). We hypothesize that activation of the adaptive hypoxic response by inhibition of PHD results in neuroprotection and prevention of vascular leakage. Mice, subjected to cerebral ischemia, were pre- or post-treated with the novel PHD inhibitor FG-4497. Inhibition of PHD activity resulted in HIF-1α stabilization, increased expression of VEGF and Epo, improved outcome from ischemic stroke and reduced edema formation by maintaining BBB integrity. Additional in vitro studies using brain endothelial cells and primary astrocytes confirmed that FG-4497 induces the HIF signaling pathway, leading to increased VEGF and Epo expression. In an in vitro ischemia model, using combined oxygen and glucose deprivation, FG-4497 promoted the survival of neurons. Furthermore, FG-4497 prevented the ischemia-induced rearrangement and gap formation of the tight junction proteins zonula occludens 1 and occludin, both in cultured endothelial cells and in infarcted brain tissue in vivo. These results indicate that FG-4497 has the potential to prevent cerebral ischemic damage by neuroprotection and prevention of vascular leakage.
Conflict of interest statement
Figures
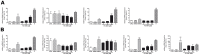
References
-
- Dirnagl U, Iadecola C, Moskowitz MA (1999) Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22: 391–397. - PubMed
-
- Gelderblom M, Leypoldt F, Steinbach K, Behrens D, Choe CU, et al. (2009) Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke 40: 1849–1857. - PubMed
-
- Sandoval KE, Witt KA (2008) Blood-brain barrier tight junction permeability and ischemic stroke. Neurobiol Dis 32: 200–219. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

