Optical and SPION-enhanced MR imaging shows that trans-stilbene inhibitors of NF-κB concomitantly lower Alzheimer's disease plaque formation and microglial activation in AβPP/PS-1 transgenic mouse brain
- PMID: 24413613
- PMCID: PMC4407807
- DOI: 10.3233/JAD-131031
Optical and SPION-enhanced MR imaging shows that trans-stilbene inhibitors of NF-κB concomitantly lower Alzheimer's disease plaque formation and microglial activation in AβPP/PS-1 transgenic mouse brain
Abstract
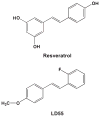
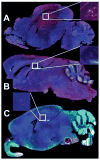
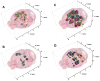
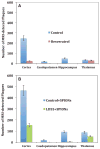
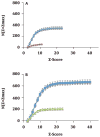
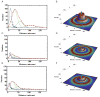
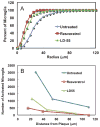
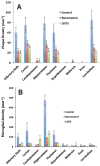
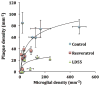
Alzheimer's disease (AD) is associated with a microglia-dependent neuroinflammatory response against plaques containing the fibrous protein amyloid-β (Aβ). Activation of microglia, which closely associate with Aβ plaques, engenders the release of pro-inflammatory cytokines and the internalization of Aβ fibrils. Since the pro-inflammatory transcription factor NF-κB is one of the major regulators of Aβ-induced inflammation, we treated transgenic amyloid-β protein protein/presenilin-1 (AβPP/PS1) mice for one year with a low dose (0.01% by weight in the diet) of either of two trans-stilbene NF-κB inhibitors, resveratrol or a synthetic analog LD55. The 3D distribution of Aβ plaques was measured ex vivo in intact brains at 60 μm resolution by quantitative magnetic resonance imaging (MRI) using blood-brain barrier-permeable, anti-AβPP-conjugated superparamagentic iron oxide nanoparticles (SPIONs). The MRI measurements were confirmed by optical microscopy of thioflavin-stained brain tissue sections and indicated that supplementation with either of the two trans-stilbenes lowered Aβ plaque density in the cortex, caudoputamen, and hippocampus by 1.4 to 2-fold. The optical measurements also included the hippocampus and indicated that resveratrol and LD55 reduced average Aβ plaque density by 2.3-fold and 3.1-fold, respectively. Ex vivo measurements of the regional distribution of microglial activation by Iba-1 immunofluorescence of brain tissue sections showed that resveratrol and LD55 reduced average microglial activation by 4.2- fold and 3.5-fold, respectively. Since LD55 lacked hydroxyl groups but both resveratrol and LD55 concomitantly reduced both Aβ plaque burden and neuroinflammation to a similar extent, it appears that the antioxidant potential of resveratrol is not an important factor in plaque reduction.
Keywords: LD55; NF-κB; SPIONs; magnetic resonance imaging; microglia; neuroinflammation; resveratrol; transgenic mice.
Figures


References
-
- Alzheimer’s Association. Alzheimer’s disease facts and figures. Alzheimer’s Association; Washington, D.C: 2011. http://www.alz.org/downloads/facts_figures_2011.pdf. - PubMed
-
- Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA. Alzheimer’s disease in the U.S. population: Prevalence estimates using the 2000 census. Arch Neurol. 2003;60:1119–1122. - PubMed
-
- Fodero-Tavoletti AT, Villemagn VL, Rowe CC, Masters CL, Barnham KJ, Cappai R. Amyloid-β: The seeds of darkness. Int J Biochem Cell Biol. 2011;43:1247–1251. - PubMed
-
- McLean CA, Cherny RA, Fraser FW, Fuller SJ, Smith MJ, Beyreuther K, Bush AI, Masters CL. Soluble pool of Abeta amyloid as a determinant of severity of neurodegeneration in Alzheimer’s disease. Ann Neurol. 1999;46:860–866. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

