A quantitative comparison of the behavior of human ventricular cardiac electrophysiology models in tissue
- PMID: 24416228
- PMCID: PMC3885549
- DOI: 10.1371/journal.pone.0084401
A quantitative comparison of the behavior of human ventricular cardiac electrophysiology models in tissue
Abstract
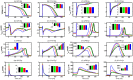
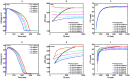
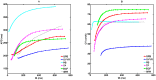
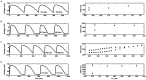
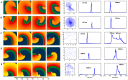
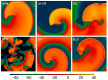
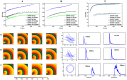
Numerical integration of mathematical models of heart cell electrophysiology provides an important computational tool for studying cardiac arrhythmias, but the abundance of available models complicates selecting an appropriate model. We study the behavior of two recently published models of human ventricular action potentials, the Grandi-Pasqualini-Bers (GPB) and the O'Hara-Virág-Varró-Rudy (OVVR) models, and compare the results with four previously published models and with available experimental and clinical data. We find the shapes and durations of action potentials and calcium transients differ between the GPB and OVVR models, as do the magnitudes and rate-dependent properties of transmembrane currents and the calcium transient. Differences also occur in the steady-state and S1-S2 action potential duration and conduction velocity restitution curves, including a maximum conduction velocity for the OVVR model roughly half that of the GPB model and well below clinical values. Between single cells and tissue, both models exhibit differences in properties, including maximum upstroke velocity, action potential amplitude, and minimum diastolic interval. Compared to experimental data, action potential durations for the GPB and OVVR models agree fairly well (although OVVR epicardial action potentials are shorter), but maximum slopes of steady-state restitution curves are smaller. Although studies show alternans in normal hearts, it occurs only in the OVVR model, and only for a narrow range of cycle lengths. We find initiated spiral waves do not progress to sustained breakup for either model. The dominant spiral wave period of the GPB model falls within clinically relevant values for ventricular tachycardia (VT), but for the OVVR model, the dominant period is longer than periods associated with VT. Our results should facilitate choosing a model to match properties of interest in human cardiac tissue and to replicate arrhythmia behavior more closely. Furthermore, by indicating areas where existing models disagree, our findings suggest avenues for further experimental work.
Conflict of interest statement
Figures



Similar articles
-
Representing variability and transmural differences in a model of human heart failure.IEEE J Biomed Health Inform. 2015 Jul;19(4):1308-20. doi: 10.1109/JBHI.2015.2442833. Epub 2015 Jun 9. IEEE J Biomed Health Inform. 2015. PMID: 26068919
-
Electrophysiological properties under heart failure conditions in a human ventricular cell: a modeling study.Annu Int Conf IEEE Eng Med Biol Soc. 2014;2014:4324-9. doi: 10.1109/EMBC.2014.6944581. Annu Int Conf IEEE Eng Med Biol Soc. 2014. PMID: 25570949
-
A computational model of pig ventricular cardiomyocyte electrophysiology and calcium handling: Translation from pig to human electrophysiology.PLoS Comput Biol. 2021 Jun 30;17(6):e1009137. doi: 10.1371/journal.pcbi.1009137. eCollection 2021 Jun. PLoS Comput Biol. 2021. PMID: 34191797 Free PMC article.
-
Developing a novel comprehensive framework for the investigation of cellular and whole heart electrophysiology in the in situ human heart: historical perspectives, current progress and future prospects.Prog Biophys Mol Biol. 2014 Aug;115(2-3):252-60. doi: 10.1016/j.pbiomolbio.2014.06.004. Epub 2014 Jun 24. Prog Biophys Mol Biol. 2014. PMID: 24972083 Review.
-
Experimental electrophysiology and arrhythmogenicity. Anisotropy and ventricular tachycardia.Eur Heart J. 1989 Sep;10 Suppl E:2-8. doi: 10.1093/eurheartj/10.suppl_e.2. Eur Heart J. 1989. PMID: 2680500 Review.
Cited by
-
Calibration of ionic and cellular cardiac electrophysiology models.Wiley Interdiscip Rev Syst Biol Med. 2020 Jul;12(4):e1482. doi: 10.1002/wsbm.1482. Epub 2020 Feb 21. Wiley Interdiscip Rev Syst Biol Med. 2020. PMID: 32084308 Free PMC article. Review.
-
Human cardiac systems electrophysiology and arrhythmogenesis: iteration of experiment and computation.Europace. 2014 Nov;16 Suppl 4(Suppl 4):iv77-iv85. doi: 10.1093/europace/euu264. Europace. 2014. PMID: 25362174 Free PMC article. Review.
-
The transient outward potassium current plays a key role in spiral wave breakup in ventricular tissue.Am J Physiol Heart Circ Physiol. 2021 Feb 1;320(2):H826-H837. doi: 10.1152/ajpheart.00608.2020. Epub 2021 Jan 1. Am J Physiol Heart Circ Physiol. 2021. PMID: 33385322 Free PMC article.
-
A mathematical model for active contraction in healthy and failing myocytes and left ventricles.PLoS One. 2017 Apr 13;12(4):e0174834. doi: 10.1371/journal.pone.0174834. eCollection 2017. PLoS One. 2017. PMID: 28406991 Free PMC article.
-
Simulation of action potential propagation based on the ghost structure method.Sci Rep. 2019 Jul 29;9(1):10927. doi: 10.1038/s41598-019-47321-2. Sci Rep. 2019. PMID: 31358816 Free PMC article.
References
-
- Fenton FH, Cherry EM (2008) Models of cardiac cell. Scholarpedia 3: 1868.
-
- Fink M, Niederer SA, Cherry EM, Fenton FH, Koivumäki JT, et al. (2011) Cardiac cell modelling: observations from the heart of the cardiac physiome project. Prog Biophys Mol Biol 104: 2–21. - PubMed
-
- Nygren A, Leon LJ, Giles WR (2001) Simulations of the human atrial action potential. Phil Trans R Soc Lond A 359: 1111–1125 10.2307/3066559 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

