Simvastatin inhibits glucose metabolism and legumain activity in human myotubes
- PMID: 24416446
- PMCID: PMC3885717
- DOI: 10.1371/journal.pone.0085721
Simvastatin inhibits glucose metabolism and legumain activity in human myotubes
Erratum in
- PLoS One. 2014;9(3):e93202
Abstract
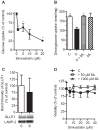
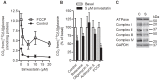
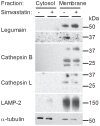
Simvastatin, a HMG-CoA reductase inhibitor, is prescribed worldwide to patients with hypercholesterolemia. Although simvastatin is well tolerated, side effects like myotoxicity are reported. The mechanism for statin-induced myotoxicity is still poorly understood. Reports have suggested impaired mitochondrial dysfunction as a contributor to the observed myotoxicity. In this regard, we wanted to study the effects of simvastatin on glucose metabolism and the activity of legumain, a cysteine protease. Legumain, being the only known asparaginyl endopeptidase, has caspase-like properties and is described to be involved in apoptosis. Recent evidences indicate a regulatory role of both glucose and statins on cysteine proteases in monocytes. Satellite cells were isolated from the Musculus obliquus internus abdominis of healthy human donors, proliferated and differentiated into polynuclear myotubes. Simvastatin with or without mevalonolactone, farnesyl pyrophosphate or geranylgeranyl pyrophosphate were introduced on day 5 of differentiation. After 48 h, cells were either harvested for immunoblotting, ELISA, cell viability assay, confocal imaging or enzyme activity analysis, or placed in a fuel handling system with [¹⁴C]glucose or [³H]deoxyglucose for uptake and oxidation studies. A dose-dependent decrease in both glucose uptake and oxidation were observed in mature myotubes after exposure to simvastatin in concentrations not influencing cell viability. In addition, simvastatin caused a decrease in maturation and activity of legumain. Dysregulation of glucose metabolism and decreased legumain activity by simvastatin points out new knowledge about the effects of statins on skeletal muscle, and may contribute to the understanding of the myotoxicity observed by statins.
Conflict of interest statement
Figures

References
-
- Strandberg TE, Pyorala K, Cook TJ, Wilhelmsen L, Faergeman O, et al. (2004) Mortality and incidence of cancer during 10-year follow-up of the Scandinavian Simvastatin Survival Study (4S). Lancet 364: 771–777. - PubMed
-
- Pedersen TR (1994) Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 344: 1383–1389. - PubMed
-
- Sinzinger H, Wolfram R, Peskar BA (2002) Muscular side effects of statins. J Cardiovasc Pharmacol 40: 163–171. - PubMed
-
- Arora R, Liebo M, Maldonado F (2006) Statin-induced myopathy: the two faces of Janus. J Cardiovasc Pharmacol Ther 11: 105–112. - PubMed
-
- Sakamoto K, Honda T, Yokoya S, Waguri S, Kimura J (2007) Rab-small GTPases are involved in fluvastatin and pravastatin-induced vacuolation in rat skeletal myofibers. FASEB J 21: 4087–4094. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

