Single Voxel Proton Spectroscopy for Neurofeedback at 7 Tesla
- PMID: 24416473
- PMCID: PMC3886242
- DOI: 10.3390/ma4091548
Single Voxel Proton Spectroscopy for Neurofeedback at 7 Tesla
Erratum in
-
Correction: Koush, Y.; Elliott, M.A. and Mathiak, K. Single Voxel Proton Spectroscopy for Neurofeedback at 7 Tesla. Materials 2011, 4, 1548-1563.Materials (Basel). 2011 Nov 24;4(11):2057-2060. doi: 10.3390/ma4112057. Materials (Basel). 2011. PMID: 28824123 Free PMC article.
Abstract
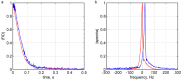
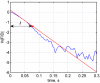
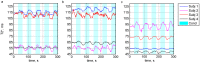
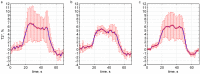
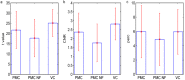
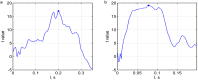
Echo-planar imaging (EPI) in fMRI is regularly used to reveal BOLD activation in presubscribed regions of interest (ROI). The response is mediated by relative changes in T2* which appear as changes in the image pixel intensities. We have proposed an application of functional single-voxel proton spectroscopy (fSVPS) for real-time studies at ultra-high MR field which can be comparable to the EPI BOLD fMRI technique. A spin-echo SVPS protocol without water suppression was acquired with 310 repetitions on a 7T Siemens MR scanner (TE/TR = 20/1000 ms, flip angle α = 90°, voxel size 10 × 10 × 10 mm3). Transmitter reference voltage was optimized for the voxel location. Spectral processing of the water signal free induction decay (FID) using log-linear regression was used to estimate the T2* change between rest and activation of a functional task. The FID spectrum was filtered with a Gaussian window around the water peak, and log-linear regression was optimized for the particular ROI by adoption of the linearization length. The spectroscopic voxel was positioned on an ROI defined from a real-time fMRI EPI BOLD localizer. Additional online signal processing algorithms performed signal drift removal (exponential moving average), despiking and low-pass filtering (modified Kalman filter) and, finally, the dynamic feedback signal normalization. Two functional tasks were used to estimate the sensitivity of the SVPS method compared to BOLD signal changes, namely the primary motor cortex (PMC, left hand finger tapping) and visual cortex (VC, blinking checkerboard). Four healthy volunteers performed these tasks and an additional session using real-time signal feedback modulating their activation level of the PMC. Results show that single voxel spectroscopy is able to provide a good and reliable estimation of the BOLD signal changes. Small data size and FID signal processing instead of processing entire brain volumes as well as more information revealed from the acquired total water spectrum, i.e., direct estimation of the T2* values and B0 changes, make SVPS proton spectroscopy suitable and advantageous for real-time neurofeedback studies. Particular challenges of ultra-high field spectroscopy due to the non-linearity in the spectral information, e.g., poor main magnetic field homogeneity and the absence of motion correction for the SVPS sequence may lead to the special artifacts in the control signal which still need to be addressed. The contrast to noise ratio (CNR), experimental statistic (t-values) and percent signal change were used as quality parameters to estimate the method performance. The potential and challenges of the spectroscopic approach for fMRI studies needs to be further investigated.
Keywords: imaging; neurofeedback; signal processing; spectroscopy.
Figures
Similar articles
-
Comparison of real-time water proton spectroscopy and echo-planar imaging sensitivity to the BOLD effect at 3 T and at 7 T.PLoS One. 2014 Mar 10;9(3):e91620. doi: 10.1371/journal.pone.0091620. eCollection 2014. PLoS One. 2014. PMID: 24614912 Free PMC article.
-
Real-time automated spectral assessment of the BOLD response for neurofeedback at 3 and 7T.J Neurosci Methods. 2013 Sep 15;218(2):148-60. doi: 10.1016/j.jneumeth.2013.05.002. Epub 2013 May 15. J Neurosci Methods. 2013. PMID: 23685226
-
Signal quality and Bayesian signal processing in neurofeedback based on real-time fMRI.Neuroimage. 2012 Jan 2;59(1):478-89. doi: 10.1016/j.neuroimage.2011.07.076. Epub 2011 Aug 4. Neuroimage. 2012. PMID: 21839842
-
On the acquisition of the water signal during water suppression: High-speed MR spectroscopic imaging with water referencing and concurrent functional MRI.NMR Biomed. 2021 May;34(5):e4261. doi: 10.1002/nbm.4261. Epub 2020 Jan 30. NMR Biomed. 2021. PMID: 31999397 Free PMC article.
-
Functional magnetic resonance imaging: the basics of blood-oxygen-level dependent (BOLD) imaging.Can Assoc Radiol J. 1998 Oct;49(5):320-9. Can Assoc Radiol J. 1998. PMID: 9803232 Review.
Cited by
-
No time for drifting: Comparing performance and applicability of signal detrending algorithms for real-time fMRI.Neuroimage. 2019 May 1;191:421-429. doi: 10.1016/j.neuroimage.2019.02.058. Epub 2019 Feb 25. Neuroimage. 2019. PMID: 30818024 Free PMC article.
-
Comparison of real-time water proton spectroscopy and echo-planar imaging sensitivity to the BOLD effect at 3 T and at 7 T.PLoS One. 2014 Mar 10;9(3):e91620. doi: 10.1371/journal.pone.0091620. eCollection 2014. PLoS One. 2014. PMID: 24614912 Free PMC article.
-
Human brain functional MRS reveals interplay of metabolites implicated in neurotransmission and neuroenergetics.J Cereb Blood Flow Metab. 2022 Jun;42(6):911-934. doi: 10.1177/0271678X221076570. Epub 2022 Jan 26. J Cereb Blood Flow Metab. 2022. PMID: 35078383 Free PMC article.
-
Real-time fMRI neurofeedback: progress and challenges.Neuroimage. 2013 Aug 1;76:386-99. doi: 10.1016/j.neuroimage.2013.03.033. Epub 2013 Mar 27. Neuroimage. 2013. PMID: 23541800 Free PMC article. Review.
-
Real-Time Semi-Automated and Automated Voxel Placement using fMRI Targets for Repeated Acquisition Magnetic Resonance Spectroscopy.J Neurosci Methods. 2023 May 15;392:109853. doi: 10.1016/j.jneumeth.2023.109853. Epub 2023 Apr 7. J Neurosci Methods. 2023. PMID: 37031764 Free PMC article.
References
-
- Goebel R. Cortex-based real-time fMRI. Neuroimage. 2001;13:S129. doi: 10.1016/S1053-8119(01)91472-7. - DOI
-
- Weiskopf N., Veit R., Erb M., Mathiak K., Grodd W., Goebel R., Birbaumer N. Physiological self-regulation of regional brain activity using real-time functional magnetic resonance imaging (fMRI): Methodology and exemplary data. Neuroimage. 2003;19:577–586. doi: 10.1016/S1053-8119(03)00145-9. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
