Ten-year effects of the advanced cognitive training for independent and vital elderly cognitive training trial on cognition and everyday functioning in older adults
- PMID: 24417410
- PMCID: PMC4055506
- DOI: 10.1111/jgs.12607
Ten-year effects of the advanced cognitive training for independent and vital elderly cognitive training trial on cognition and everyday functioning in older adults
Abstract
Objectives: To determine the effects of cognitive training on cognitive abilities and everyday function over 10 years.
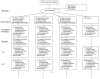
Design: Ten-year follow-up of a randomized, controlled single-blind trial (Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE)) with three intervention groups and a no-contact control group.
Setting: Six U.S. cities.
Participants: A volunteer sample of 2,832 persons (mean baseline age 73.6; 26% African American) living independently.
Intervention: Ten training sessions for memory, reasoning, or speed of processing; four sessions of booster training 11 and 35 months after initial training.
Measurements: Objectively measured cognitive abilities and self-reported and performance-based measures of everyday function.
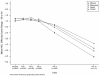
Results: Participants in each intervention group reported less difficulty with instrumental activities of daily living (IADLs) (memory: effect size = 0.48, 99% confidence interval (CI) = 0.12-0.84; reasoning: effect size = 0.38, 99% CI = 0.02-0.74; speed of processing: effect size = 0.36, 99% CI = 0.01-0.72). At a mean age of 82, approximately 60% of trained participants, versus 50% of controls (P < .05), were at or above their baseline level of self-reported IADL function at 10 years. The reasoning and speed-of-processing interventions maintained their effects on their targeted cognitive abilities at 10 years (reasoning: effect size = 0.23, 99% CI = 0.09-0.38; speed of processing: effect size = 0.66, 99% CI = 0.43-0.88). Memory training effects were no longer maintained for memory performance. Booster training produced additional and durable improvement for the reasoning intervention for reasoning performance (effect size = 0.21, 99% CI = 0.01-0.41) and the speed-of-processing intervention for speed-of-processing performance (effect size = 0.62, 99% CI = 0.31-0.93).
Conclusion: Each Advanced Cognitive Training for Independent and Vital Elderly cognitive intervention resulted in less decline in self-reported IADL compared with the control group. Reasoning and speed, but not memory, training resulted in improved targeted cognitive abilities for 10 years.
Keywords: cognitive abilities; cognitive training; elderly; everyday function; training maintenance.
© 2014, Copyright the Authors Journal compilation © 2014, The American Geriatrics Society.
Figures


References
-
- Hertzog C, Kramer A, Wilson R, et al. Enrichment effects on adult cognitive development: can the functional capacity of older adults be preserved and enhanced? Psychol Sci. 2008;9(1):1–65. - PubMed
-
- Rebok G. State-of-Science Review: SR:E22 UK. Government Foresight Mental Capital and Mental Wellbeing Project. Government Office for Science; 2008. Cognitive training: influence on neuropsychological and brain function in later life.
-
- Pappa K, Walsh S, Snyder P. Immediate and delayed effects of cognitive interventions in healthy elderly: a review of current literature and future directions. Alzheimers Dement. 2009;5(1):50–60. - PubMed
Publication types
MeSH terms
Grants and funding
- U01AG-14260/AG/NIA NIH HHS/United States
- U01 AG14263/AG/NIA NIH HHS/United States
- U01 NR004508/NR/NINR NIH HHS/United States
- U19 AG010483/AG/NIA NIH HHS/United States
- U01 AG014282/AG/NIA NIH HHS/United States
- U01 AG014260/AG/NIA NIH HHS/United States
- U01NR04508/NR/NINR NIH HHS/United States
- U01 AG14289/AG/NIA NIH HHS/United States
- R24 HD042828/HD/NICHD NIH HHS/United States
- R03 AG023078/AG/NIA NIH HHS/United States
- U01AG14276/AG/NIA NIH HHS/United States
- U01 AG014276/AG/NIA NIH HHS/United States
- P30 AG028740/AG/NIA NIH HHS/United States
- U01 AG014263/AG/NIA NIH HHS/United States
- U01 AG014289/AG/NIA NIH HHS/United States
- U01 AG14282/AG/NIA NIH HHS/United States
- U01NR04507/NR/NINR NIH HHS/United States
- U01 NR004507/NR/NINR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

