Crystallization and preliminary X-ray study of Vibrio cholerae uridine phosphorylase in complex with 6-methyluracil
- PMID: 24419619
- PMCID: PMC3943090
- DOI: 10.1107/S2053230X13031877
Crystallization and preliminary X-ray study of Vibrio cholerae uridine phosphorylase in complex with 6-methyluracil
Abstract
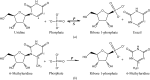
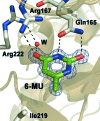
Uridine phosphorylase catalyzes the phosphorolysis of ribonucleosides, with the nitrogenous base and ribose 1-phosphate as products. Additionally, it catalyzes the reverse reaction of the synthesis of ribonucleosides from ribose 1-phosphate and a nitrogenous base. However, the enzyme does not catalyze the synthesis of nucleosides when the substrate is a nitrogenous base substituted at the 6-position, such as 6-methyluracil (6-MU). In order to explain this fact, it is essential to investigate the three-dimensional structure of the complex of 6-MU with uridine phosphorylase. 6-MU is a pharmaceutical agent that improves tissue nutrition and enhances cell regeneration by normalization of nucleotide exchange in humans. 6-MU is used for the treatment of diseases of the gastrointestinal tract, including infectious diseases. Here, procedures to obtain the uridine phosphorylase from the pathogenic bacterium Vibrio cholerae (VchUPh), purification of this enzyme, crystallization of the complex of VchUPh with 6-MU, and X-ray data collection and preliminary X-ray analysis of the VchUPh-6-MU complex at atomic resolution are reported.
Keywords: 6-methyluracil; Vibrio cholerae; bacterial uridine phosphorylase.
Figures

Similar articles
-
Expression, purification, crystallization and preliminary X-ray structure analysis of Vibrio cholerae uridine phosphorylase in complex with thymidine.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Nov 1;68(Pt 11):1394-7. doi: 10.1107/S1744309112041401. Epub 2012 Oct 30. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012. PMID: 23143257 Free PMC article.
-
X-ray structure of Salmonella typhimurium uridine phosphorylase complexed with 5-fluorouracil and molecular modelling of the complex of 5-fluorouracil with uridine phosphorylase from Vibrio cholerae.Acta Crystallogr D Biol Crystallogr. 2012 Aug;68(Pt 8):968-74. doi: 10.1107/S090744491201815X. Epub 2012 Jul 17. Acta Crystallogr D Biol Crystallogr. 2012. PMID: 22868762
-
Crystallization of uridine phosphorylase from Shewanella oneidensis MR-1 in the laboratory and under microgravity and preliminary X-ray diffraction analysis.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Nov 1;68(Pt 11):1387-9. doi: 10.1107/S1744309112041784. Epub 2012 Oct 30. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012. PMID: 23143255 Free PMC article.
-
Development of inhibitors of pyrimidine metabolism.Yonsei Med J. 1989 Dec;30(4):315-26. doi: 10.3349/ymj.1989.30.4.315. Yonsei Med J. 1989. PMID: 2697111 Review.
-
Uridine phosophorylase: an important enzyme in pyrimidine metabolism and fluoropyrimidine activation.Drugs Today (Barc). 2004 May;40(5):431-43. doi: 10.1358/dot.2004.40.5.850491. Drugs Today (Barc). 2004. PMID: 15319798 Review.
References
-
- Caradoc-Davies, T. T., Cutfield, S. M., Lamont, I. L. & Cutfield, J. F. (2004). J. Mol. Biol. 337, 337–354. - PubMed
-
- Chadaev, A. P. & Klimiashvili, A. D. (2003). Khirurgiia (Mosk.), pp. 54–56. - PubMed
-
- DeLano, W. L. (2002). PyMOL http://www.pymol.org.
-
- el-On, J. & Weinrauch, L. (1990). Int. J. Dermatol. 29, 232. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

