Polyoxometalates active against tumors, viruses, and bacteria
- PMID: 24420711
- PMCID: PMC7122307
- DOI: 10.1007/978-3-642-41004-8_4
Polyoxometalates active against tumors, viruses, and bacteria
Abstract
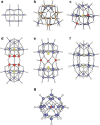
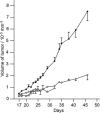
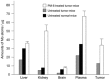
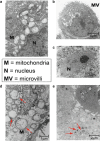
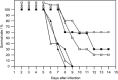
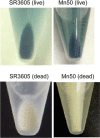
Polyoxometalates (PMs) as discrete metal-oxide cluster anions with high solubility in water and photochemically and electrochemically active property have a wide variety of structures not only in molecular size from sub-nano to sub-micrometers with a various combination of metals but also in symmetry and highly negative charge. One of the reasons for such a structural variety originates from their conformation change (due to the condensed aggregation and the structural assembly) which strongly depends on environmental parameters such as solution pH, concentration, and coexistent foreign inorganic and/or organic substances. In the course of the application of the physicochemical properties of such PMs to the medical fields, antitumoral, antiviral, and antibacterial activities have been developed for realization of a novel inorganic medicine which provides a biologically excellent activity never replaced by other approved medicines. Several PMs as a candidate for clinical uses have been licensed toward the chemotherapy of solid tumors (such as human gastric cancer and pancreatic cancer), DNA and RNA viruses (such as HSV, HIV, influenza, and SARS), and drug-resistant bacteria (such as MRSA and VRSA) in recent years: [NH3Pr(i)]6[Mo7O24]∙3H2O (PM-8) and [Me3NH]6[H2Mo(V) 12O28(OH)12(Mo(VI)O3)4]∙2H2O (PM-17) for solid tumors; K7[PTi2W10O40]∙6H2O (PM-19), [Pr(i)NH3]6H[PTi2W10O38(O2)2]∙H2O (PM-523), and K11H[(VO)3(SbW9O33)2]∙27H2O (PM-1002) for viruses; and K6[P2W18O62]∙14H2O (PM-27), K4[SiMo12O40]∙3H2O (SiMo12), and PM-19 for MRSA and VRSA. The results are discussed from a point of view of the chemotherapeutic clarification in this review.
Figures















References
-
- Baba M, Snoeck R, Pauwels R, DeClerq E. Sulfated polysaccharides are potent and selective inhibitors of various enveloped viruses including herpes simplex viruses, cytomegalovirus, vesicular stomatitis viruses, and human immunodeficiency virus. Antimicrob Agents Chemother. 1988;32:1742–1745. - PMC - PubMed
-
- Barnard DL, Hill CL, Gage T, Matheson JE, Huffmann JH, Sidwell RW, Otto MI, Schinazi RF. Potent inhibition of respiratory syncytial virus by polyoxometalates of several structure classes. Antiviral Res. 1997;34:27–37. - PubMed
-
- Blumenthal R, Clgue MJ, Durell SR, Epand RM. Membrane fusion. Chem Rev. 2003;103:53–69. - PubMed
-
- Boneca IG, Chiosis G. Vancomycin resistance: occurrence, mechanism and strategies to combat it. Expert Opin Ther Targets. 2003;7:311–328. - PubMed
-
- Borrás-Alamenar JJ, Coronado E, Müller A, Pope MT, editors. Polyoxometalate molecular science. Dordrecht: Kluwer; 2003.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

