Stromal fibroblasts mediate extracellular matrix remodeling and invasion of scirrhous gastric carcinoma cells
- PMID: 24427313
- PMCID: PMC3888433
- DOI: 10.1371/journal.pone.0085485
Stromal fibroblasts mediate extracellular matrix remodeling and invasion of scirrhous gastric carcinoma cells
Abstract
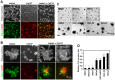
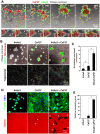
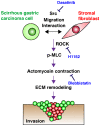
Scirrhous gastric carcinoma (SGC) has the worst prognosis of all gastric cancers, owing to its rapid expansion by invasion and frequent peritoneal dissemination. Due to the increased proliferation of stromal fibroblasts (SFs) that occurs within SGC lesions and the peritoneal metastatic sites, SFs have been proposed to support the progression of this disease. However, the biological and molecular basis and the pathological role of the intercellular interaction between SGC cells and SFs remain largely unknown. In this study, we investigated the role of SFs in the invasion of the extracellular matrix (ECM) by SGC cells. When SGC cells were cocultured with SFs derived from SGC tissue on three-dimensional (3D) Matrigel, they were attracted together to form large cellular aggregates that invaded within the Matrigel. Time-lapse imaging revealed that this process was associated with extensive contraction and remodeling of the ECM. Immunofluorescence and biochemical analysis showed that SGC cells stimulate phosphorylation of myosin light chain and actomyosin-mediated mechanical remodeling of the ECM by SFs. By utilizing this assay system for inhibitor library screening, we have identified several inhibitors that potently suppress the cooperation between SGC cells and SFs to form the invasive structures. Among them, a Src inhibitor dasatinib impaired the interaction between SGC cells and SFs both in vitro and in vivo and effectively blocked peritoneal dissemination of SGC cells. These results indicate that SFs mediate mechanical remodeling of the ECM by SGC cells, thereby promoting invasion and peritoneal dissemination of SGC.
Conflict of interest statement
Figures




References
-
- Calvo F, Sahai E (2011) Cell communication networks in cancer invasion. Curr Opin Cell Biol 23: 621–629. - PubMed
-
- Kalluri R, Zeisberg M (2006) Fibroblasts in cancer. Nat Rev Cancer 6: 392–401. - PubMed
-
- De Wever O, Demetter P, Mareel M, Bracke M (2008) Stromal myofibroblasts are drivers of invasive cancer growth. Int J Cancer 123: 2229–2238. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

