Targeting Wnt signaling at the neuroimmune interface for dopaminergic neuroprotection/repair in Parkinson's disease
- PMID: 24431301
- PMCID: PMC4061726
- DOI: 10.1093/jmcb/mjt053
Targeting Wnt signaling at the neuroimmune interface for dopaminergic neuroprotection/repair in Parkinson's disease
Abstract
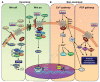
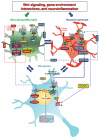
During the past three decades, the Wingless-type MMTV integration site (Wnt) signaling cascade has emerged as an essential system regulating multiple processes in developing and adult brain. Accumulating evidence points to a dysregulation of Wnt signaling in major neurodegenerative pathologies including Parkinson's disease (PD), a common neurodegenerative disorder characterized by the progressive loss of midbrain dopaminergic (mDA) neurons and deregulated activation of astrocytes and microglia. This review highlights the emerging link between Wnt signaling and key inflammatory pathways during mDA neuron damage/repair in PD progression. In particular, we summarize recent evidence documenting that aging and neurotoxicant exposure strongly antagonize Wnt/β-catenin signaling in mDA neurons and subventricular zone (SVZ) neuroprogenitors via astrocyte-microglial interactions. Dysregulation of the crosstalk between Wnt/β-catenin signaling and anti-oxidant/anti-inflammatory pathways delineate novel mechanisms driving the decline of SVZ plasticity with age and the limited nigrostriatal dopaminergic self-repair in PD. These findings hold a promise in developing therapies that target Wnt/β-catenin signaling to enhance endogenous restoration and neuronal outcome in age-dependent diseases, such as PD.
Keywords: Parkinson's disease; Wnt/β-catenin signaling; dopaminergic neurons; neurodegeneration; neurogenesis; neuroinflammation; neuroprotection.
Figures

References
-
- Abou-Sleiman PM, Muqit MM, Wood NW. Expanding insights of mitochondrial dysfunction in Parkinson’s disease. Nat. Rev. Neurosci. 2006;7:207–219. - PubMed
-
- Adachi K, Mirzadeh Z, Sakaguchi M, et al. β-catenin signaling promotes proliferation of progenitor cells in the adult mouse subventricular zone. Stem Cells. 2007;25:2827–2836. - PubMed
-
- Aloisi F. The role of microglia and astrocytes in CNS immune surveillance and immunopathology. Adv. Exp. Med. Biol. 1999;468:123–133. - PubMed
-
- Angers S, Moon RT. Proximal events in Wnt signal transduction. Nat. Rev. Mol. Cell Biol. 2009;10:468–477. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

