Dopamine deficiency contributes to early visual dysfunction in a rodent model of type 1 diabetes
- PMID: 24431431
- PMCID: PMC3891954
- DOI: 10.1523/JNEUROSCI.3483-13.2014
Dopamine deficiency contributes to early visual dysfunction in a rodent model of type 1 diabetes
Abstract
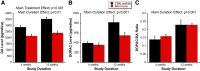
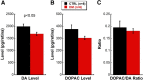
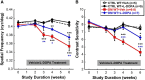
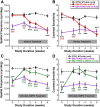
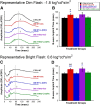
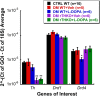
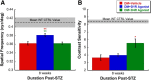
Dopamine (DA) functions as an essential neuromodulator in the brain and retina such that disruptions in the dopaminergic system are associated with common neurologic disorders such as Parkinson's disease. Although a reduction in DA content has been observed in diabetes, its effects in the development of diabetes-induced neuropathy remains unknown. Because the retina is rich in DA and has a well known diabetes-induced pathology (diabetic retinopathy or DR), this study was designed to examine the role of retinal DA deficiency in early visual defects in DR. Using rodent models of type 1 diabetes mellitus, we investigated whether diabetes caused a reduction in retinal DA content in both rats and mice and determined whether restoring DA levels or activating specific DA receptor pathways could improve visual function (evaluated with optokinetic tracking response) of diabetic mice, potentially via improvement of retinal function (assessed with electroretinography). We found that diabetes significantly reduced DA levels by 4 weeks in rats and by 5 weeks in mice, coincident with the initial detection of visual deficits. Treatment with l-DOPA, a DA precursor, improved overall retinal and visual functions in diabetic mice and acute treatment with DA D1 or D4 receptor agonists improved spatial frequency threshold or contrast sensitivity, respectively. Together, our results indicate that retinal DA deficiency is an underlying mechanism for early, diabetes-induced visual dysfunction and suggest that therapies targeting the retinal dopaminergic system may be beneficial in early-stage DR.
Keywords: Diabetic retinopathy; Dopamine; Visual function.
Figures

Similar articles
-
Dopamine Deficiency Mediates Early Rod-Driven Inner Retinal Dysfunction in Diabetic Mice.Invest Ophthalmol Vis Sci. 2018 Jan 1;59(1):572-581. doi: 10.1167/iovs.17-22692. Invest Ophthalmol Vis Sci. 2018. PMID: 29372256 Free PMC article.
-
Neuroprotective effects of PPARα in retinopathy of type 1 diabetes.PLoS One. 2019 Feb 4;14(2):e0208399. doi: 10.1371/journal.pone.0208399. eCollection 2019. PLoS One. 2019. PMID: 30716067 Free PMC article.
-
TrkB signalling pathway mediates the protective effects of exercise in the diabetic rat retina.Eur J Neurosci. 2018 May;47(10):1254-1265. doi: 10.1111/ejn.13909. Epub 2018 Apr 3. Eur J Neurosci. 2018. PMID: 29537701 Free PMC article.
-
[Preretinopathic changes in the oscillatory potential in diabetic retina: interpretation and significance].Nippon Ganka Gakkai Zasshi. 1998 Dec;102(12):813-36. Nippon Ganka Gakkai Zasshi. 1998. PMID: 10025114 Review. Japanese.
-
Somatostatin and diabetic retinopathy: current concepts and new therapeutic perspectives.Endocrine. 2014 Jun;46(2):209-14. doi: 10.1007/s12020-014-0232-z. Epub 2014 Mar 14. Endocrine. 2014. PMID: 24627166 Review.
Cited by
-
Diabetic retinopathy alters light-induced clock gene expression and dopamine levels in the mouse retina.Mol Vis. 2016 Aug 5;22:959-69. eCollection 2016. Mol Vis. 2016. PMID: 27559292 Free PMC article.
-
A systems view of type 2 diabetes-associated metabolic perturbations in saliva, blood and urine at different timescales of glycaemic control.Diabetologia. 2015 Aug;58(8):1855-67. doi: 10.1007/s00125-015-3636-2. Epub 2015 Jun 7. Diabetologia. 2015. PMID: 26049400 Free PMC article.
-
Acute Visual Impairment in a Patient with Parkinson's Disease after Successful Bilateral Subthalamic Nucleus Deep Brain Stimulation with Low-Dose Levodopa: A Case Report.Brain Sci. 2023 Jan 5;13(1):103. doi: 10.3390/brainsci13010103. Brain Sci. 2023. PMID: 36672084 Free PMC article.
-
Retinal Neurodegeneration as an Early Manifestation of Diabetic Eye Disease and Potential Neuroprotective Therapies.Curr Diab Rep. 2019 Feb 26;19(4):17. doi: 10.1007/s11892-019-1134-5. Curr Diab Rep. 2019. PMID: 30806815 Free PMC article. Review.
-
Inhibition of phosphorylated calcium/calmodulin-dependent protein kinase IIα relieves streptozotocin-induced diabetic neuropathic pain through regulation of P2X3 receptor in dorsal root ganglia.Purinergic Signal. 2023 Mar;19(1):99-111. doi: 10.1007/s11302-021-09829-z. Epub 2022 Jan 1. Purinergic Signal. 2023. PMID: 34973115 Free PMC article.
References
-
- Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, Kester M, Kimball SR, Krady JK, LaNoue KF, Norbury CC, Quinn PG, Sandirasegarane L, Simpson IA, Simpson IA. Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes. 2006;55:2401–2411. doi: 10.2337/db05-1635. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
