Subcellular expression and neuroprotective effects of SK channels in human dopaminergic neurons
- PMID: 24434522
- PMCID: PMC4040692
- DOI: 10.1038/cddis.2013.530
Subcellular expression and neuroprotective effects of SK channels in human dopaminergic neurons
Abstract
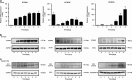
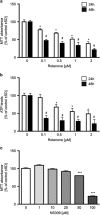
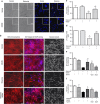
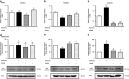
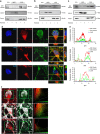
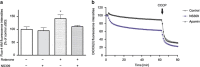
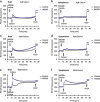
Small-conductance Ca(2+)-activated K(+) channel activation is an emerging therapeutic approach for treatment of neurological diseases, including stroke, amyotrophic lateral sclerosis and schizophrenia. Our previous studies showed that activation of SK channels exerted neuroprotective effects through inhibition of NMDAR-mediated excitotoxicity. In this study, we tested the therapeutic potential of SK channel activation of NS309 (25 μM) in cultured human postmitotic dopaminergic neurons in vitro conditionally immortalized and differentiated from human fetal mesencephalic cells. Quantitative RT-PCR and western blotting analysis showed that differentiated dopaminergic neurons expressed low levels of SK2 channels and high levels of SK1 and SK3 channels. Further, protein analysis of subcellular fractions revealed expression of SK2 channel subtype in mitochondrial-enriched fraction. Mitochondrial complex I inhibitor rotenone (0.5 μM) disrupted the dendritic network of human dopaminergic neurons and induced neuronal death. SK channel activation reduced mitochondrial membrane potential, while it preserved the dendritic network, cell viability and ATP levels after rotenone challenge. Mitochondrial dysfunction and delayed dopaminergic cell death were prevented by increasing and/or stabilizing SK channel activity. Overall, our findings show that activation of SK channels provides protective effects in human dopaminergic neurons, likely via activation of both membrane and mitochondrial SK channels. Thus, SK channels are promising therapeutic targets for neurodegenerative disorders such as Parkinson's disease, where dopaminergic cell loss is associated with progression of the disease.
Figures
Similar articles
-
Mitochondrial small conductance SK2 channels prevent glutamate-induced oxytosis and mitochondrial dysfunction.J Biol Chem. 2013 Apr 12;288(15):10792-804. doi: 10.1074/jbc.M113.453522. Epub 2013 Feb 19. J Biol Chem. 2013. PMID: 23430260 Free PMC article.
-
NS309 decreases rat detrusor smooth muscle membrane potential and phasic contractions by activating SK3 channels.Br J Pharmacol. 2013 Apr;168(7):1611-25. doi: 10.1111/bph.12049. Br J Pharmacol. 2013. PMID: 23145946 Free PMC article.
-
SK2 and SK3 expression differentially affect firing frequency and precision in dopamine neurons.Neuroscience. 2012 Aug 16;217:67-76. doi: 10.1016/j.neuroscience.2012.04.053. Epub 2012 Apr 30. Neuroscience. 2012. PMID: 22554781 Free PMC article.
-
Modulation of the activity of dopaminergic neurons by SK channels: a potential target for the treatment of Parkinson's disease?Neurosci Bull. 2010 Jun;26(3):265-71. doi: 10.1007/s12264-010-1217-4. Neurosci Bull. 2010. PMID: 20502506 Free PMC article. Review.
-
Modulation of small conductance calcium-activated potassium (SK) channels: a new challenge in medicinal chemistry.Curr Med Chem. 2003 Apr;10(8):625-47. doi: 10.2174/0929867033457908. Curr Med Chem. 2003. PMID: 12678783 Review.
Cited by
-
Differential modulation of SK channel subtypes by phosphorylation.Cell Calcium. 2021 Mar;94:102346. doi: 10.1016/j.ceca.2020.102346. Epub 2021 Jan 6. Cell Calcium. 2021. PMID: 33422768 Free PMC article.
-
Targeting the Small- and Intermediate-Conductance Ca-Activated Potassium Channels: The Drug-Binding Pocket at the Channel/Calmodulin Interface.Neurosignals. 2014;22(2):65-78. doi: 10.1159/000367896. Epub 2014 Oct 8. Neurosignals. 2014. PMID: 25300231 Free PMC article. Review.
-
SK2 channels regulate mitochondrial respiration and mitochondrial Ca2+ uptake.Cell Death Differ. 2017 May;24(5):761-773. doi: 10.1038/cdd.2017.2. Epub 2017 Mar 10. Cell Death Differ. 2017. PMID: 28282037 Free PMC article.
-
VTA dopamine neurons are hyperexcitable in 3xTg-AD mice due to casein kinase 2-dependent SK channel dysfunction.bioRxiv [Preprint]. 2023 Nov 17:2023.11.16.567486. doi: 10.1101/2023.11.16.567486. bioRxiv. 2023. Update in: Nat Commun. 2024 Nov 8;15(1):9673. doi: 10.1038/s41467-024-53891-1. PMID: 38014232 Free PMC article. Updated. Preprint.
-
Activation of SK/KCa Channel Attenuates Spinal Cord Ischemia-Reperfusion Injury via Anti-oxidative Activity and Inhibition of Mitochondrial Dysfunction in Rabbits.Front Pharmacol. 2019 Apr 2;10:325. doi: 10.3389/fphar.2019.00325. eCollection 2019. Front Pharmacol. 2019. PMID: 31001121 Free PMC article.
References
-
- Wang Y, Yang PL, Tang JF, Lin JF, Cai XH, Wang XT, et al. Potassium channels: possible new therapeutic targets in Parkinson's disease. Med Hypotheses. 2008;71:546–550. - PubMed
-
- Wang S, Hu LF, Yang Y, Ding JH, Hu G. Studies of ATP-sensitive potassium channels on 6-hydroxydopamine and haloperidol rat models of Parkinson's disease: implications for treating Parkinson's disease. Neuropharmacology. 2005;48:984–992. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

