Proteomic and functional consequences of hexokinase deficiency in glucose-repressible Kluyveromyces lactis
- PMID: 24434903
- PMCID: PMC3945914
- DOI: 10.1074/mcp.M113.032714
Proteomic and functional consequences of hexokinase deficiency in glucose-repressible Kluyveromyces lactis
Abstract
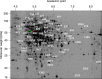
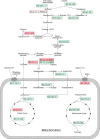
The analysis of glucose signaling in the Crabtree-positive eukaryotic model organism Saccharomyces cerevisiae has disclosed a dual role of its hexokinase ScHxk2, which acts as a glycolytic enzyme and key signal transducer adapting central metabolism to glucose availability. In order to identify evolutionarily conserved characteristics of hexokinase structure and function, the cellular response of the Crabtree-negative yeast Kluyveromyces lactis to rag5 null mutation and concomitant deficiency of its unique hexokinase KlHxk1 was analyzed by means of difference gel electrophoresis. In total, 2,851 fluorescent spots containing different protein species were detected in the master gel representing all of the K. lactis proteins that were solubilized from glucose-grown KlHxk1 wild-type and mutant cells. Mass spectrometric peptide analysis identified 45 individual hexokinase-dependent proteins related to carbohydrate, short-chain fatty acid and tricarboxylic acid metabolism as well as to amino acid and protein turnover, but also to general stress response and chromatin remodeling, which occurred as a consequence of KlHxk1 deficiency at a minimum 3-fold enhanced or reduced level in the mutant proteome. In addition, three proteins exhibiting homology to 2-methylcitrate cycle enzymes of S. cerevisiae were detected at increased concentrations, suggesting a stimulation of pyruvate formation from amino acids and/or fatty acids. Experimental validation of the difference gel electrophoresis approach by post-lysis dimethyl labeling largely confirmed the abundance changes detected in the mutant proteome via the former method. Taking into consideration the high proportion of identified hexokinase-dependent proteins exhibiting increased proteomic levels, KlHxk1 is likely to have a repressive function in a multitude of metabolic pathways. The proteomic alterations detected in the mutant classify KlHxk1 as a multifunctional enzyme and support the view of evolutionary conservation of dual-role hexokinases even in organisms that are less specialized than S. cerevisiae in terms of glucose utilization.
Figures

Similar articles
-
Protein kinase Ymr291w/Tda1 is essential for glucose signaling in saccharomyces cerevisiae on the level of hexokinase isoenzyme ScHxk2 phosphorylation*.J Biol Chem. 2015 Mar 6;290(10):6243-55. doi: 10.1074/jbc.M114.595074. Epub 2015 Jan 15. J Biol Chem. 2015. PMID: 25593311 Free PMC article.
-
The unique hexokinase of Kluyveromyces lactis. Molecular and functional characterization and evaluation of a role in glucose signaling.J Biol Chem. 2003 Oct 10;278(41):39280-6. doi: 10.1074/jbc.M305706200. Epub 2003 Jul 25. J Biol Chem. 2003. PMID: 12882981
-
Crystal structure of hexokinase KlHxk1 of Kluyveromyces lactis: a molecular basis for understanding the control of yeast hexokinase functions via covalent modification and oligomerization.J Biol Chem. 2010 Dec 24;285(52):41019-33. doi: 10.1074/jbc.M110.185850. Epub 2010 Oct 12. J Biol Chem. 2010. PMID: 20943665 Free PMC article.
-
In vivo phosphorylation and in vitro autophosphorylation-inactivation of Kluyveromyces lactis hexokinase KlHxk1.Biochem Biophys Res Commun. 2013 May 31;435(2):313-8. doi: 10.1016/j.bbrc.2013.03.121. Epub 2013 Apr 10. Biochem Biophys Res Commun. 2013. PMID: 23583397
-
Yeast Interspecies Comparative Proteomics Reveals Divergence in Expression Profiles and Provides Insights into Proteome Resource Allocation and Evolutionary Roles of Gene Duplication.Mol Cell Proteomics. 2016 Jan;15(1):218-35. doi: 10.1074/mcp.M115.051854. Epub 2015 Nov 11. Mol Cell Proteomics. 2016. PMID: 26560065 Free PMC article.
Cited by
-
Protein kinase Ymr291w/Tda1 is essential for glucose signaling in saccharomyces cerevisiae on the level of hexokinase isoenzyme ScHxk2 phosphorylation*.J Biol Chem. 2015 Mar 6;290(10):6243-55. doi: 10.1074/jbc.M114.595074. Epub 2015 Jan 15. J Biol Chem. 2015. PMID: 25593311 Free PMC article.
-
Hexokinase and Glucokinases Are Essential for Fitness and Virulence in the Pathogenic Yeast Candida albicans.Front Microbiol. 2019 Feb 25;10:327. doi: 10.3389/fmicb.2019.00327. eCollection 2019. Front Microbiol. 2019. PMID: 30858840 Free PMC article.
References
-
- Flores C. L., Rodriguez C., Petit T., Gancedo C. (2000) Carbohydrate and energy-yielding metabolism in non-conventional yeasts. FEMS Microbiol. Rev. 24, 507–529 - PubMed
-
- Rolland F., Winderickx J., Thevelein J. M. (2001) Glucose-sensing mechanisms in eukaryotic cells. Trends Biochem. Sci. 26, 310–317 - PubMed
-
- Matschinsky F. M. (2009) Assessing the potential of glucokinase activators in diabetes therapy. Nat. Rev. Drug Discov. 8, 399–416 - PubMed
-
- Rolland F., Baena-Gonzalez E., Sheen J. (2006) Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu. Rev. Plant Biol. 57, 675–709 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

