Morphology, projection pattern, and neurochemical identity of Cajal's "centrifugal neurons": the cells of origin of the tectoventrogeniculate pathway in pigeon (Columba livia) and chicken (Gallus gallus)
- PMID: 24435811
- PMCID: PMC4011500
- DOI: 10.1002/cne.23539
Morphology, projection pattern, and neurochemical identity of Cajal's "centrifugal neurons": the cells of origin of the tectoventrogeniculate pathway in pigeon (Columba livia) and chicken (Gallus gallus)
Abstract
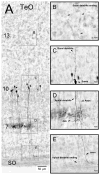
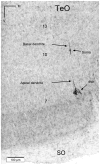
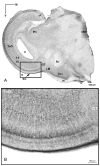
The nucleus geniculatus lateralis pars ventralis (GLv) is a prominent retinal target in all amniotes. In birds, it is in receipt of a dense and topographically organized retinal projection. The GLv is also the target of substantial and topographically organized projections from the optic tectum and the visual wulst (hyperpallium). Tectal and retinal afferents terminate homotopically within the external GLv-neuropil. Efferents from the GLv follow a descending course through the tegmentum and can be traced into the medial pontine nucleus. At present, the cells of origin of the Tecto-GLv projection are only partially described. Here we characterized the laminar location, morphology, projection pattern, and neurochemical identity of these cells by means of neural tracer injections and intracellular fillings in slice preparations and extracellular tracer injections in vivo. The Tecto-GLv projection arises from a distinct subset of layer 10 bipolar neurons, whose apical dendrites show a complex transverse arborization at the level of layer 7. Axons of these bipolar cells arise from the apical dendrites and follow a course through the optic tract to finally form very fine and restricted terminal endings inside the GLv-neuropil. Double-label experiments showed that these bipolar cells were choline acetyltransferase (ChAT)-immunoreactive. Our results strongly suggest that Tecto-GLv neurons form a pathway by which integrated tectal activity rapidly feeds back to the GLv and exerts a focal cholinergic modulation of incoming retinal inputs.
Keywords: ChAT; GLv; birds; optic tectum; slice; vine-neuron.
Copyright © 2014 Wiley Periodicals, Inc.
Conflict of interest statement
Figures











Similar articles
-
An immunocytochemical analysis of the lateral geniculate complex in the pigeon (Columba livia).J Comp Neurol. 1991 Dec 22;314(4):721-49. doi: 10.1002/cne.903140407. J Comp Neurol. 1991. PMID: 1687743
-
Distribution, laminar location, and morphology of tectal neurons projecting to the isthmo-optic nucleus and the nucleus isthmi, pars parvocellularis in the pigeon (Columba livia) and chick (Gallus domesticus): a retrograde labelling study.J Comp Neurol. 1991 Mar 15;305(3):470-88. doi: 10.1002/cne.903050310. J Comp Neurol. 1991. PMID: 1709956
-
Chemically specific retinal ganglion cells collateralize to the pars ventralis of the lateral geniculate nucleus and optic tectum in the pigeon (Columba livia).Vis Neurosci. 1989 Nov;3(5):477-82. doi: 10.1017/s0952523800005976. Vis Neurosci. 1989. PMID: 2484822
-
The isthmic nuclei providing parallel feedback connections to the avian tectum have different neurochemical identities: Expression of glutamatergic and cholinergic markers in the chick (Gallus gallus).J Comp Neurol. 2015 Jun 15;523(9):1341-58. doi: 10.1002/cne.23739. Epub 2015 Mar 12. J Comp Neurol. 2015. PMID: 25594665
-
Topographic projections of the retina and optic tectum upon the ventral lateral geniculate nucleus in the chick.J Comp Neurol. 1979 May 1;185(1):87-106. doi: 10.1002/cne.901850106. J Comp Neurol. 1979. PMID: 429617
Cited by
-
The Medial Ventrothalamic Circuitry: Cells Implicated in a Bimodal Network.Front Neural Circuits. 2018 Feb 9;12:9. doi: 10.3389/fncir.2018.00009. eCollection 2018. Front Neural Circuits. 2018. PMID: 29479309 Free PMC article.
-
AP-2δ Expression Kinetics in Multimodal Networks in the Developing Chicken Midbrain.Front Neural Circuits. 2021 Oct 21;15:756184. doi: 10.3389/fncir.2021.756184. eCollection 2021. Front Neural Circuits. 2021. PMID: 34744640 Free PMC article.
-
Unraveling circuits of visual perception and cognition through the superior colliculus.Neuron. 2021 Mar 17;109(6):918-937. doi: 10.1016/j.neuron.2021.01.013. Epub 2021 Feb 5. Neuron. 2021. PMID: 33548173 Free PMC article. Review.
-
A thalamic hub-and-spoke network enables visual perception during action by coordinating visuomotor dynamics.Nat Neurosci. 2025 Mar;28(3):627-639. doi: 10.1038/s41593-025-01874-w. Epub 2025 Feb 10. Nat Neurosci. 2025. PMID: 39930095 Free PMC article.
-
Visual-Cerebellar Pathways and Their Roles in the Control of Avian Flight.Front Neurosci. 2018 Apr 9;12:223. doi: 10.3389/fnins.2018.00223. eCollection 2018. Front Neurosci. 2018. PMID: 29686605 Free PMC article. Review.
References
-
- Angaut P, Repérant J. Fine structure of the optic fibre termination layers in the pigeon optic tectum: A golgi and electron microscope study. Neuroscience. 1976;1(2):93-IN13. - PubMed
-
- Atoji Y. Immunohistochemical localization of vesicular glutamate transporter 2 (vGluT2) in the central nervous system of the pigeon (Columba livia) The Journal of Comparative Neurology. 2011;519(14):2887–2905. - PubMed
-
- Bagnoli P, Fontanesi G, Alesci R, Erichsen JT. Distribution of neuropeptide Y, substance P, and Choline acetyltransferase in the developing visual system of the pigeon and effects of unilateral retina removal. The Journal of Comparative Neurology. 1992;318(4):392–414. - PubMed
-
- Batista SS, Pires RS, Britto LR. Differential expression of AMPA-type glutamate receptor subunits during development of the chick optic tectum. Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas/Sociedade Brasileira de Biofisica [et al] 2002;35(8):973–978. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

