Titania-promoted carboxylic acid alkylations of alkenes and cascade addition-cyclizations
- PMID: 24437519
- PMCID: PMC3963454
- DOI: 10.1021/jo4027929
Titania-promoted carboxylic acid alkylations of alkenes and cascade addition-cyclizations
Abstract
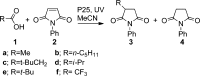
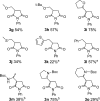
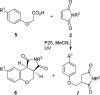
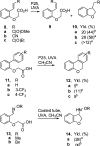
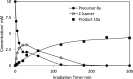
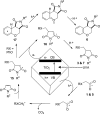
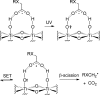
Photochemical reactions employing TiO2 and carboxylic acids under dry anaerobic conditions led to several types of C-C bond-forming processes with electron-deficient alkenes. The efficiency of alkylation varied appreciably with substituents in the carboxylic acids. The reactions of aryloxyacetic acids with maleimides resulted in a cascade process in which a pyrrolochromene derivative accompanied the alkylated succinimide. The selectivity for one or other of these products could be tuned to some extent by employing the photoredox catalyst under different conditions. Aryloxyacetic acids adapted for intramolecular ring closures by inclusion of 2-alkenyl, 2-aryl, or 2-oximinyl functionality reacted rather poorly. Profiles of reactant consumption and product formation for these systems were obtained by an in situ NMR monitoring technique. An array of different catalyst forms were tested for efficiency and ease of use. The proposed mechanism, involving hole capture at the TiO2 surface by the carboxylates followed by CO2 loss, was supported by EPR spectroscopic evidence of the intermediates. Deuterium labeling indicated that the titania likely donates protons from surface hydroxyl groups as well as supplying electrons and holes, thus acting as both a catalyst and a reaction partner.
Figures

References
-
- For a review, see:Calvert J. G.; Pitts J. N.. Photochemistry; Wiley: New York, 1966; pp 427–434.
-
- Budac D.; Wan P. J. Photochem. Photobiol., A 1992, 67, 135–166.
-
- For a review, see:Nonhebel D. C.; Walton J. C. In Free Radical Chemistry; Cambridge University Press: Cambridge, U.K., 1974; pp 99–127.
-
- Barton D. H. R.; Crich D.; Motherwell W. B. J. Chem. Soc., Chem. Commun. 1983, 939–941.
- Motherwell W. B.; Crich D.. Free Radical Chain Reactions in Organic Synthesis; Academic Press: London, 1992.
-
- Johnson R. G.; Ingham R. K. Chem. Rev. 1956, 56, 219–269.
- Baker F. W.; Holtz H. D.; Stock L. M. J. Org. Chem. 1963, 28, 514–516.
LinkOut - more resources
Full Text Sources
Other Literature Sources

