Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial
- PMID: 24439297
- PMCID: PMC4171740
- DOI: 10.1016/S0140-6736(13)62117-0
Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial
Abstract
Background: Choroideremia is an X-linked recessive disease that leads to blindness due to mutations in the CHM gene, which encodes the Rab escort protein 1 (REP1). We assessed the effects of retinal gene therapy with an adeno-associated viral (AAV) vector encoding REP1 (AAV.REP1) in patients with this disease.
Methods: In a multicentre clinical trial, six male patients (aged 35-63 years) with choroideremia were administered AAV.REP1 (0·6-1·0×10(10) genome particles, subfoveal injection). Visual function tests included best corrected visual acuity, microperimetry, and retinal sensitivity tests for comparison of baseline values with 6 months after surgery. This study is registered with ClinicalTrials.gov, number NCT01461213.
Findings: Despite undergoing retinal detachment, which normally reduces vision, two patients with advanced choroideremia who had low baseline best corrected visual acuity gained 21 letters and 11 letters (more than two and four lines of vision). Four other patients with near normal best corrected visual acuity at baseline recovered to within one to three letters. Mean gain in visual acuity overall was 3·8 letters (SE 4·1). Maximal sensitivity measured with dark-adapted microperimetry increased in the treated eyes from 23·0 dB (SE 1·1) at baseline to 25·3 dB (1·3) after treatment (increase 2·3 dB [95% CI 0·8-3·8]). In all patients, over the 6 months, the increase in retinal sensitivity in the treated eyes (mean 1·7 [SE 1·0]) was correlated with the vector dose administered per mm(2) of surviving retina (r=0·82, p=0·04). By contrast, small non-significant reductions (p>0·05) were noted in the control eyes in both maximal sensitivity (-0·8 dB [1·5]) and mean sensitivity (-1·6 dB [0·9]). One patient in whom the vector was not administered to the fovea re-established variable eccentric fixation that included the ectopic island of surviving retinal pigment epithelium that had been exposed to vector.
Interpretation: The initial results of this retinal gene therapy trial are consistent with improved rod and cone function that overcome any negative effects of retinal detachment. These findings lend support to further assessment of gene therapy in the treatment of choroideremia and other diseases, such as age-related macular degeneration, for which intervention should ideally be applied before the onset of retinal thinning.
Funding: UK Department of Health and Wellcome Trust.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures

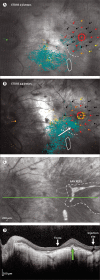

Comment in
-
Gene therapy arrives at the macula.Lancet. 2014 Mar 29;383(9923):1105-7. doi: 10.1016/S0140-6736(14)60033-7. Epub 2014 Jan 16. Lancet. 2014. PMID: 24439295 No abstract available.
-
Sensory systems: Promising results in a gene therapy trial for retinal disease.Nat Rev Neurol. 2014 Mar;10(3):123. doi: 10.1038/nrneurol.2014.18. Epub 2014 Feb 4. Nat Rev Neurol. 2014. PMID: 24492352 No abstract available.
References
-
- Mauthner L. Ein Fall von Choroideremie. Ber Naturwissensch-med Ver Inssbruck. 1872;2:191–197.
-
- Sankila EM, Tolvanen R, van den Hurk JA, Cremers FP, de la Chapelle A. Aberrant splicing of the CHM gene is a significant cause of choroideremia. Nat Genet. 1992;1:109–113. - PubMed
-
- Cremers FP, van de Pol DJ, van Kerkhoff LP, Wieringa B, Ropers HH. Cloning of a gene that is rearranged in patients with choroideraemia. Nature. 1990;347:674–677. - PubMed
-
- van Bokhoven H, van den Hurk JAJM, Bogerd LPM. Cloning and characterization of the human choroideremia gene. Hum Mol Genet. 1994;3:1041–1046. - PubMed
-
- Seabra MC, Brown MS, Goldstein JL. Retinal degeneration in choroideremia: deficiency of rab geranylgeranyl transferase. Science. 1993;259:377–381. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

