Costimulatory molecule DNAM-1 is essential for optimal differentiation of memory natural killer cells during mouse cytomegalovirus infection
- PMID: 24440149
- PMCID: PMC3943894
- DOI: 10.1016/j.immuni.2013.12.011
Costimulatory molecule DNAM-1 is essential for optimal differentiation of memory natural killer cells during mouse cytomegalovirus infection
Abstract
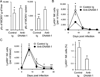
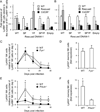
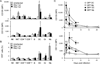
Recent studies demonstrate that natural killer (NK) cells have adaptive immune features. Here, we investigated the role of the costimulatory molecule DNAM-1 in the differentiation of NK cells in a mouse model of cytomegalovirus (MCMV) infection. Antibody blockade of DNAM-1 suppressed the expansion of MCMV-specific Ly49H(+) cells during viral infection and inhibited the generation of memory NK cells. Similarly, DNAM-1-deficient (Cd226(-/-)) Ly49H(+) NK cells exhibited intrinsic defects in expansion and differentiation into memory cells. Src-family tyrosine kinase Fyn and serine-threonine protein kinase C isoform eta (PKCη) signaling through DNAM-1 played distinct roles in the generation of MCMV-specific effector and memory NK cells. Thus, cooperative signaling through DNAM-1 and Ly49H are required for NK cell-mediated host defense against MCMV infection.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
Tracking Effector and Memory NK Cells During MCMV Infection.Methods Mol Biol. 2016;1441:1-12. doi: 10.1007/978-1-4939-3684-7_1. Methods Mol Biol. 2016. PMID: 27177652
-
Caspase-8 restricts natural killer cell accumulation during MCMV Infection.Med Microbiol Immunol. 2019 Aug;208(3-4):543-554. doi: 10.1007/s00430-019-00617-6. Epub 2019 May 21. Med Microbiol Immunol. 2019. PMID: 31115653 Free PMC article.
-
Tracking the fate of antigen-specific versus cytokine-activated natural killer cells after cytomegalovirus infection.J Exp Med. 2016 Nov 14;213(12):2745-2758. doi: 10.1084/jem.20160726. Epub 2016 Oct 24. J Exp Med. 2016. PMID: 27810928 Free PMC article.
-
Caspase-8-dependent control of NK- and T cell responses during cytomegalovirus infection.Med Microbiol Immunol. 2019 Aug;208(3-4):555-571. doi: 10.1007/s00430-019-00616-7. Epub 2019 May 16. Med Microbiol Immunol. 2019. PMID: 31098689 Review.
-
Genetic control of innate immune responses against cytomegalovirus: MCMV meets its match.Genes Immun. 2002 Aug;3(5):250-62. doi: 10.1038/sj.gene.6363876. Genes Immun. 2002. PMID: 12140743 Review.
Cited by
-
Chronic T cell receptor stimulation unmasks NK receptor signaling in peripheral T cell lymphomas via epigenetic reprogramming.J Clin Invest. 2021 Jul 1;131(13):e139675. doi: 10.1172/JCI139675. J Clin Invest. 2021. PMID: 34043588 Free PMC article.
-
Memory responses of innate lymphocytes and parallels with T cells.Semin Immunopathol. 2018 Jul;40(4):343-355. doi: 10.1007/s00281-018-0686-9. Epub 2018 May 28. Semin Immunopathol. 2018. PMID: 29808388 Free PMC article. Review.
-
Human cytomegalovirus drives epigenetic imprinting of the IFNG locus in NKG2Chi natural killer cells.PLoS Pathog. 2014 Oct 16;10(10):e1004441. doi: 10.1371/journal.ppat.1004441. eCollection 2014 Oct. PLoS Pathog. 2014. PMID: 25329659 Free PMC article.
-
Proliferative capacity exhibited by human liver-resident CD49a+CD25+ NK cells.PLoS One. 2017 Aug 9;12(8):e0182532. doi: 10.1371/journal.pone.0182532. eCollection 2017. PLoS One. 2017. PMID: 28792982 Free PMC article.
-
Cd226-/- natural killer cells fail to establish stable contacts with cancer cells and show impaired control of tumor metastasis in vivo.Oncoimmunology. 2017 Jul 5;6(8):e1338994. doi: 10.1080/2162402X.2017.1338994. eCollection 2017. Oncoimmunology. 2017. PMID: 28920003 Free PMC article.
References
-
- Aoki J, Koike S, Asou H, Ise I, Suwa H, Tanaka T, Miyasaka M, Nomoto A. Mouse homolog of poliovirus receptor-related gene 2 product, mPRR2, mediates homophilic cell aggregation. Exp Cell Res. 1997;235:374–384. - PubMed
-
- Arase H, Mocarski ES, Campbell AE, Hill AB, Lanier LL. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science. 2002;296:1323–1326. - PubMed
-
- Bakker AB, Hoek RM, Cerwenka A, Blom B, Lucian L, McNeil T, Murray R, Phillips LH, Sedgwick JD, Lanier LL. DAP12-deficient mice fail to develop autoimmunity due to impaired antigen priming. Immunity. 2000;13:345–353. - PubMed
-
- Brown MG, Dokun AO, Heusel JW, Smith HR, Beckman DL, Blattenberger EA, Dubbelde CE, Stone LR, Scalzo AA, Yokoyama WM. Vital involvement of a natural killer cell activation receptor in resistance to viral infection. Science. 2001;292:934–937. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

